TRIS, 5 kg, CAS No. 77-86-1 | Biological Buffer Reagents | Biochemistry | Life Science | Carl Roth - International

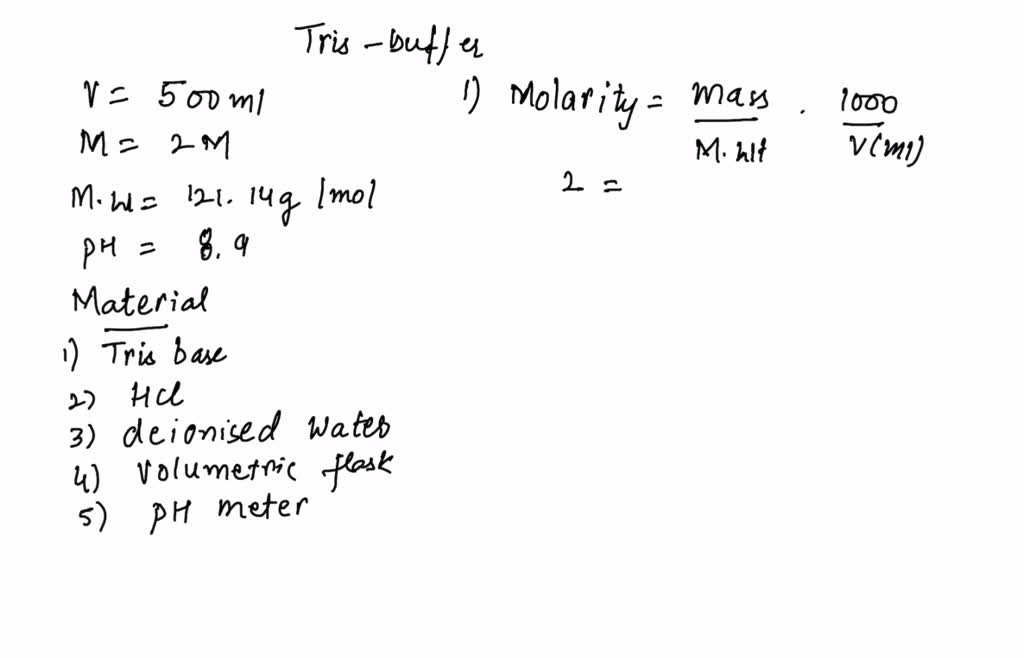
SOLVED: 1. Describe how you would make the following two buffers (remember you need to calculate the mass of Tris base and the mass of Tris-acetate needed). Please show your work. a.


![T60040-1000.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 1 Kilogram T60040-1000.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 1 Kilogram](https://d2gdaxkudte5p.cloudfront.net/system/images/T60040-1000.0_.jpg)







![Tris Base [C4H11NO3] Molecular Weight Calculation - Laboratory Notes Tris Base [C4H11NO3] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2022/11/tris-base-molecular-weight-calculation-300x204.jpg)





![T60040-500.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 500 Grams T60040-500.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 500 Grams](https://d2gdaxkudte5p.cloudfront.net/system/images/T60040-500.0_.jpg)