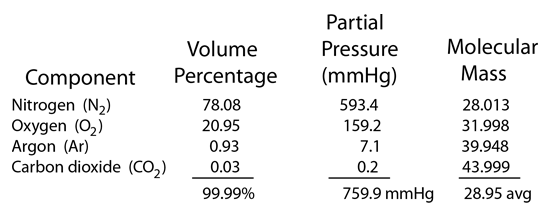
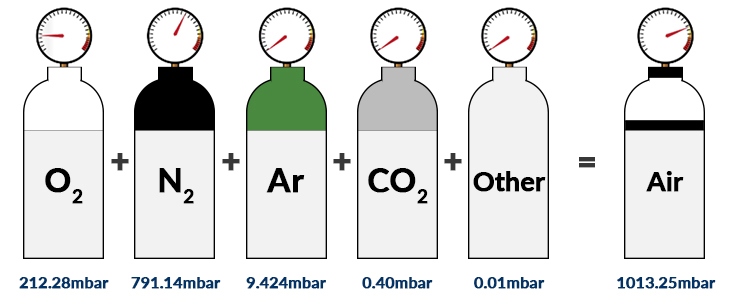
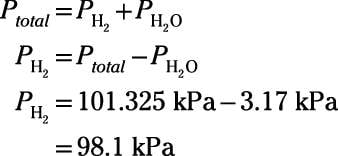Worked example: Calculating the equilibrium total pressure after a change in volume | Khan Academy - YouTube
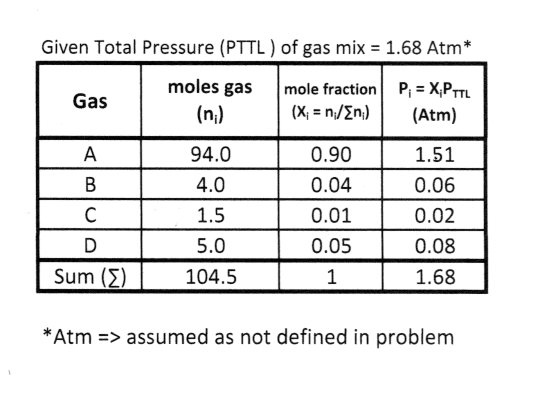
Assuming that the total pressure of the gases is 1.68 and that their mole ratio is 94 : 4.0 : 1.5 : 0.50, how do you calculate the partial pressure (in atmospheres) of each gas? | Socratic