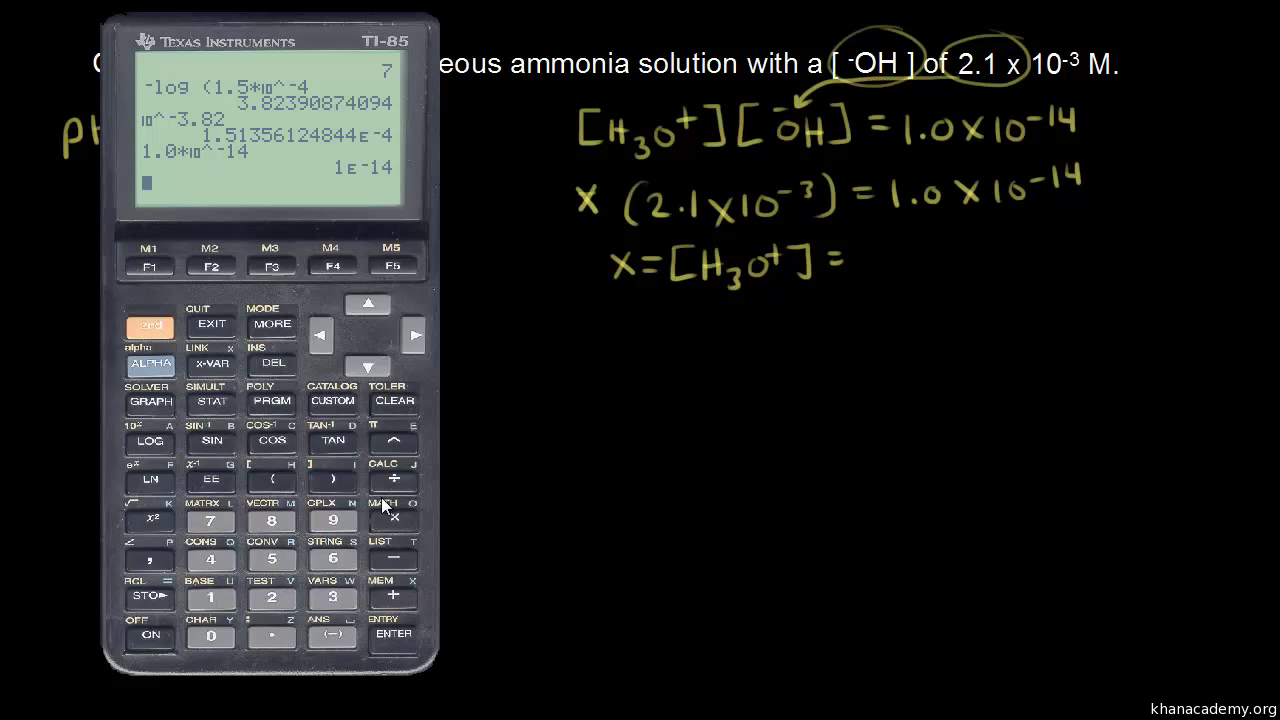
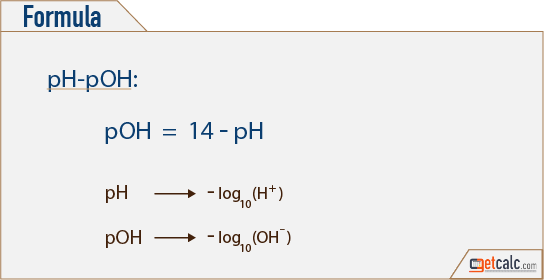
The pH of a solution describes its acidity and is the negative logarithm ( log) of its hydrogen ion concentration. The term pH is used because the hydrogen ion concentration in solutions of weak acids and in many other fluids is frequently much less than 1 ...
Would a solution with a [H+] concentration of 1 X 10-11 have a pH value of ___ and be considered a(n)? - Quora

![Finding the pH, pOH, [H+], [OH-] - ACIDS AND BASES: IT'S ACTUALLY Quite "BASIC" Finding the pH, pOH, [H+], [OH-] - ACIDS AND BASES: IT'S ACTUALLY Quite "BASIC"](http://itsactuallyquitebasic.weebly.com/uploads/2/7/8/0/27808159/4950515.png?357)


![Solving For pH, pOH, [H+], [OH-] - Acids & Bases Solving For pH, pOH, [H+], [OH-] - Acids & Bases](http://youarebasic.weebly.com/uploads/5/0/1/4/50143245/5802360_orig.png)







![Given [H+] or [OH-], Calculate pH & pOH - YouTube Given [H+] or [OH-], Calculate pH & pOH - YouTube](https://i.ytimg.com/vi/ghIYaqo0Ycc/maxresdefault.jpg)

![pH = -log[H+] pH = -log[H+]](https://s3.studylib.net/store/data/008427242_1-6a856521ca6b3bcc442fa00c77f4fe6e.png)
![Слайд 24 pH of Common Substances Слайд 25 Calculating the pH pH = - log [H+] (Remember that the [ Слайд 24 pH of Common Substances Слайд 25 Calculating the pH pH = - log [H+] (Remember that the [](https://www.sliderbase.com/images/referats/1143b/(25).PNG)




![Calculations of pH, pOH, [H+] and [OH-] Calculations of pH, pOH, [H+] and [OH-]](https://www.sciencegeek.net/Chemistry/taters/graphics/pHSchematic.gif)