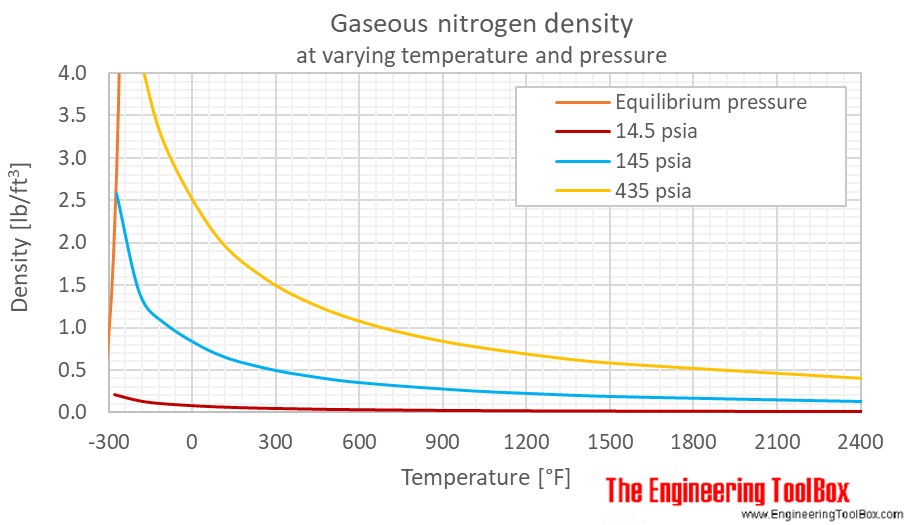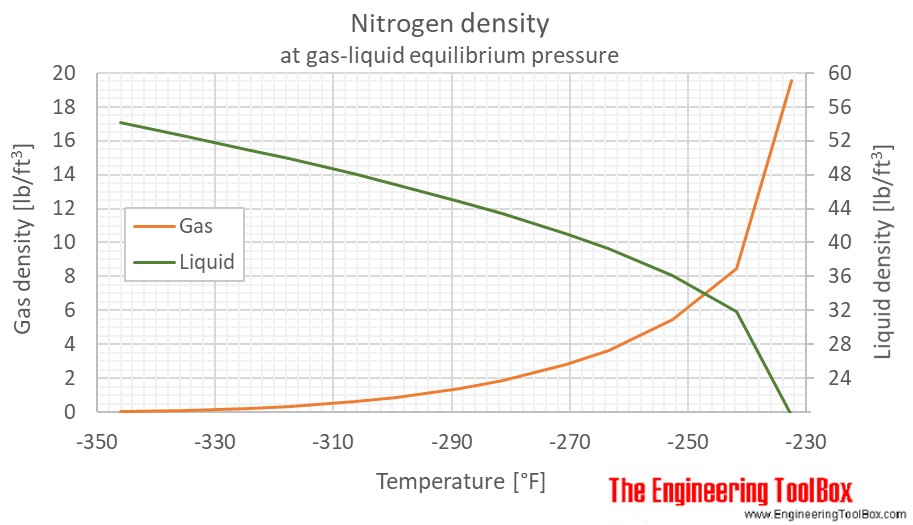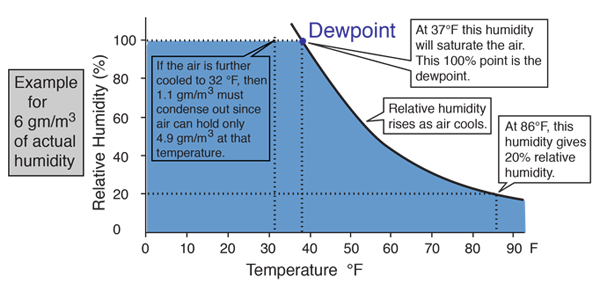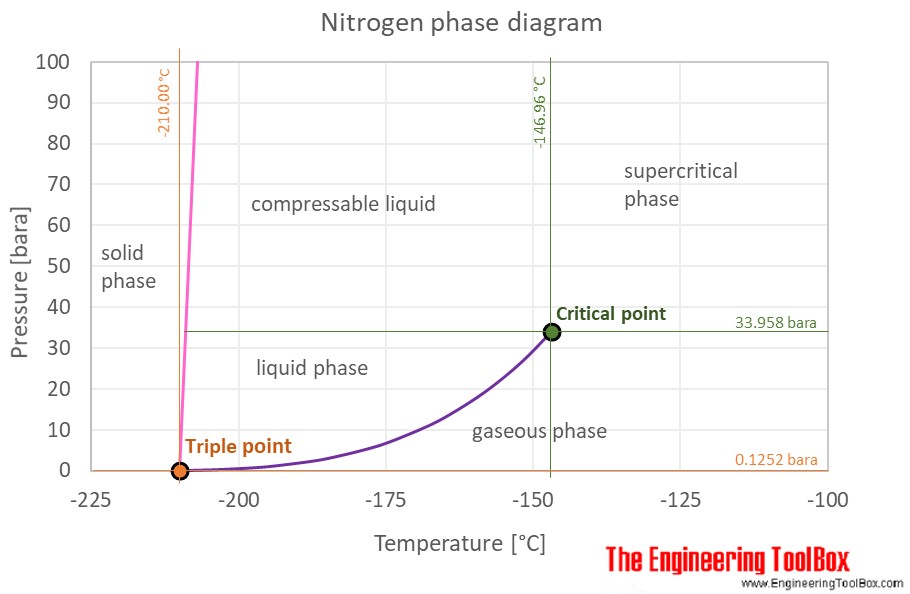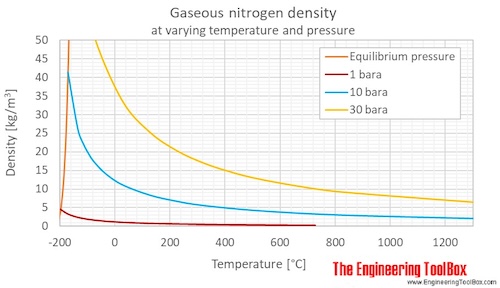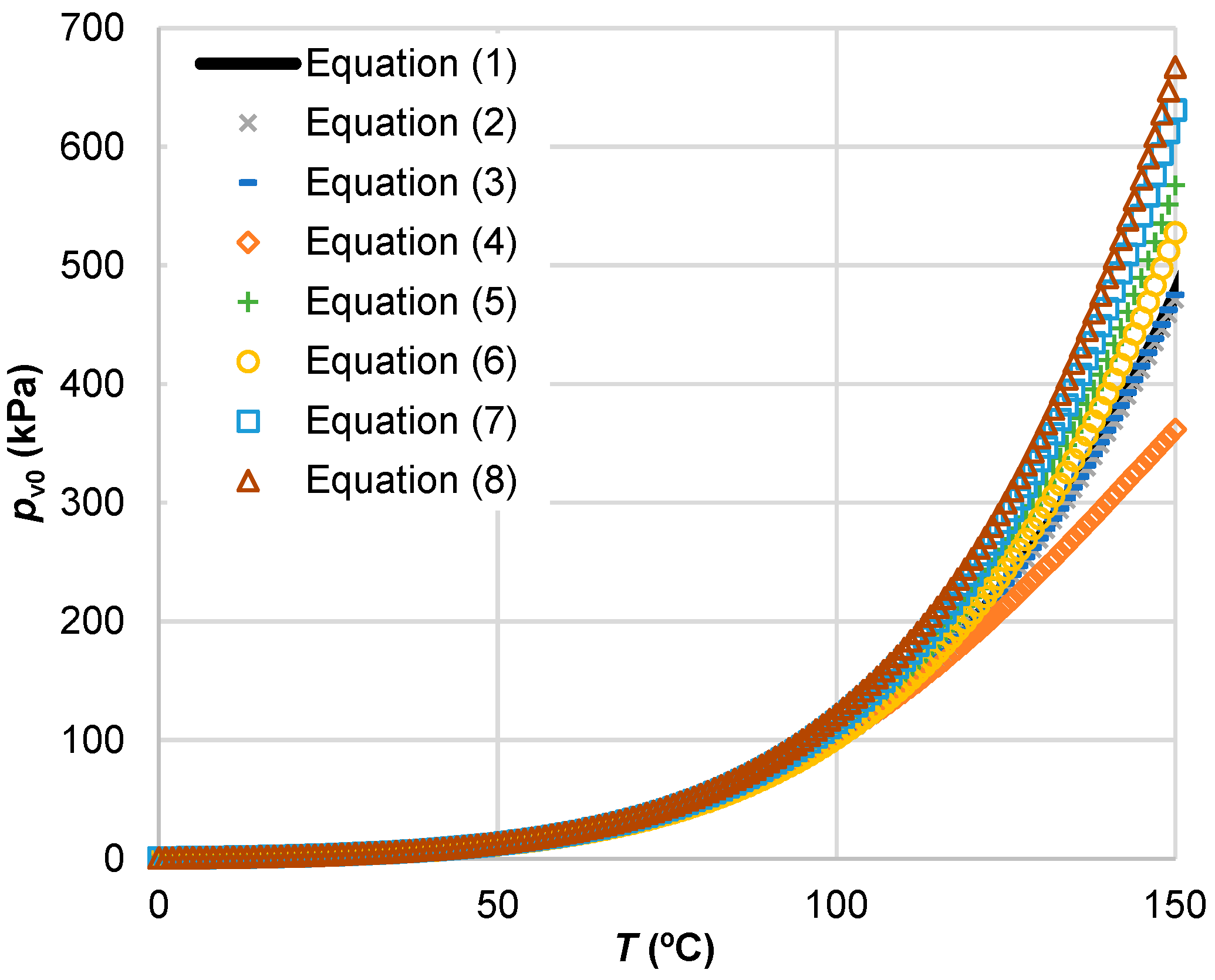
Applied Sciences | Free Full-Text | Consistency of Water Vapour Pressure and Specific Heat Capacity Values for Modelling Clay-Based Engineered Barriers

HTS conductor temperature and liquid nitrogen pressure distributions... | Download Scientific Diagram
ACP - Reappraising the appropriate calculation of a common meteorological quantity: potential temperature

Liquid nitrogen has a boiling point of `-195.81^@C` at atmospheric pressure. Calculate this - YouTube