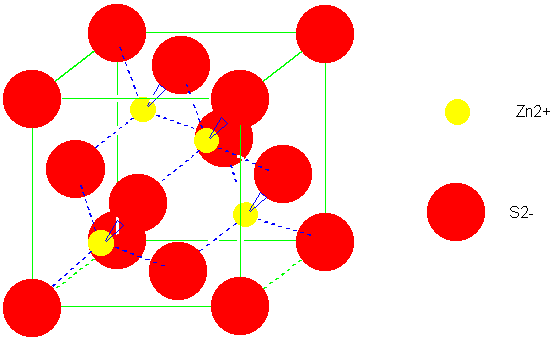
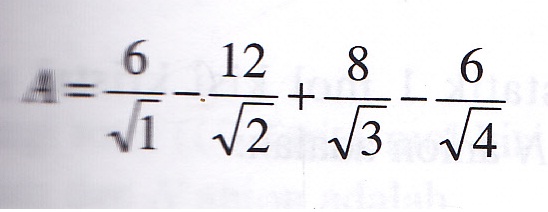The calculation of the Madelung constant of a “generalized” sodium chloride lattice - Transactions of the Faraday Society (RSC Publishing)

The calculation of the madelung constant for NaCl structure using the method of images - Nafari - 1987 - physica status solidi (a) - Wiley Online Library
Show that the Madelung constant for a one-dimensional array of ions of alternating sign with - Sarthaks eConnect | Largest Online Education Community
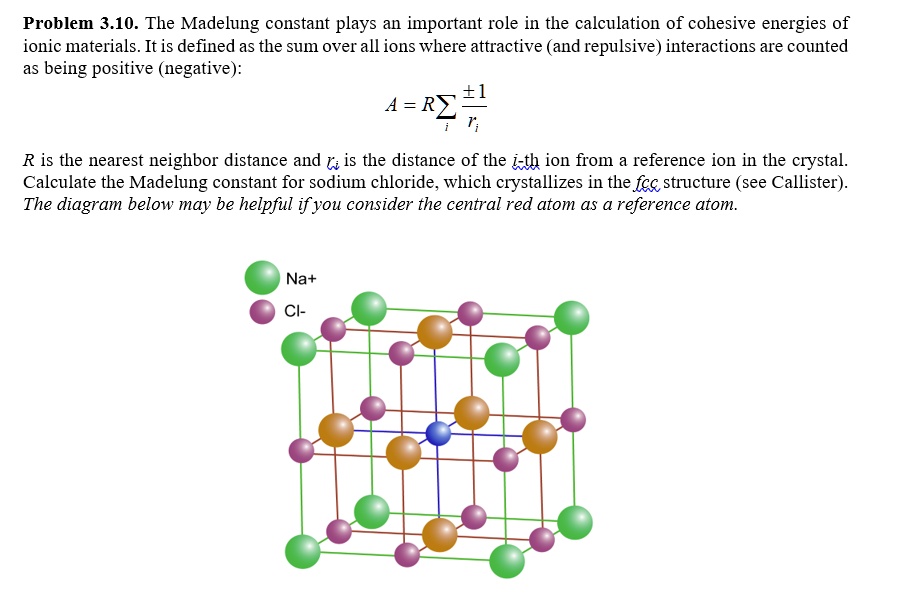
SOLVED: Problem 3.10. The Madelung constant plays an important role in the calculation of cohesive energies of ionic materials. It is defined as the sum over all ions where attractive (and repulsive)

solid state physics - Madelung constant for a two dimensional square crystal lattice - Physics Stack Exchange