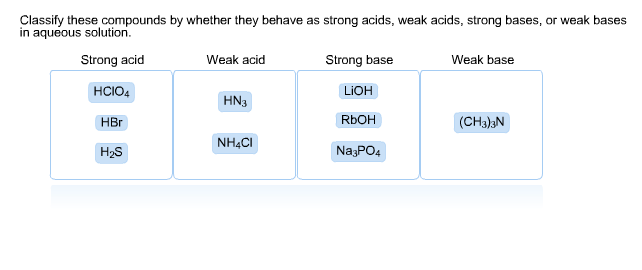
SOLVED: Predict the products of the following acidlbase reactions: LiOH (aq) HBrkoq) LiOH (aq) HBr(aq) noreaction LiOH (aq) HBrkaq) H,Od) + LiBr(aq) LiOH (aq) HBrkag) Li (aq) + OH" (aq) LiOH(aq) +

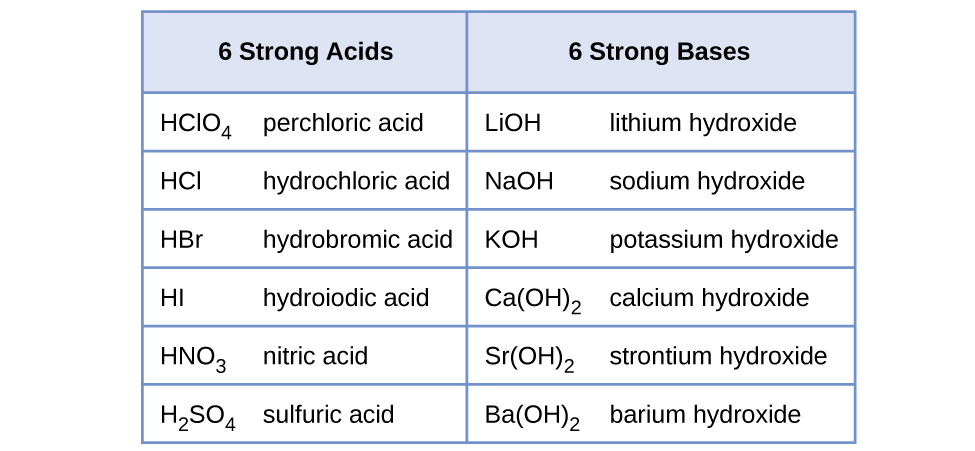
PPT - STRONG BASES LiOH, NaOH, KOH, RbOH, CsOH, Ca(OH) 2 , Sr(OH) 2 , Ba(OH) 2 PowerPoint Presentation - ID:4763546

Equilibrium – Acids and Bases. Review of Acids and Bases Arrhenius Theory of Acids and Bases ▫An acid is a substance that dissociates in water to produce. - ppt download

Question Video: Calculating the Concentration of Lithium Hydroxide in Millimolars Via Titration with Sulfuric Acid | Nagwa

SOLVED:Name each of the following acids or bases: a. H3 PO4 b. LiOH c. HI d. HCN e. HCIO2 f. Ca(OH)2