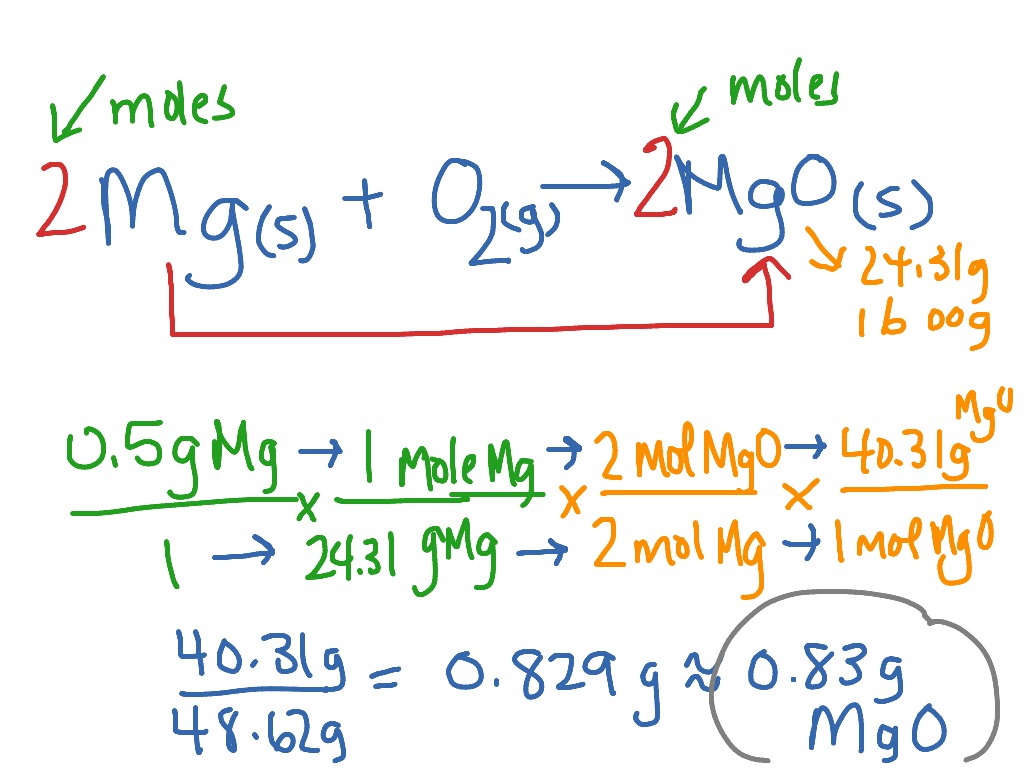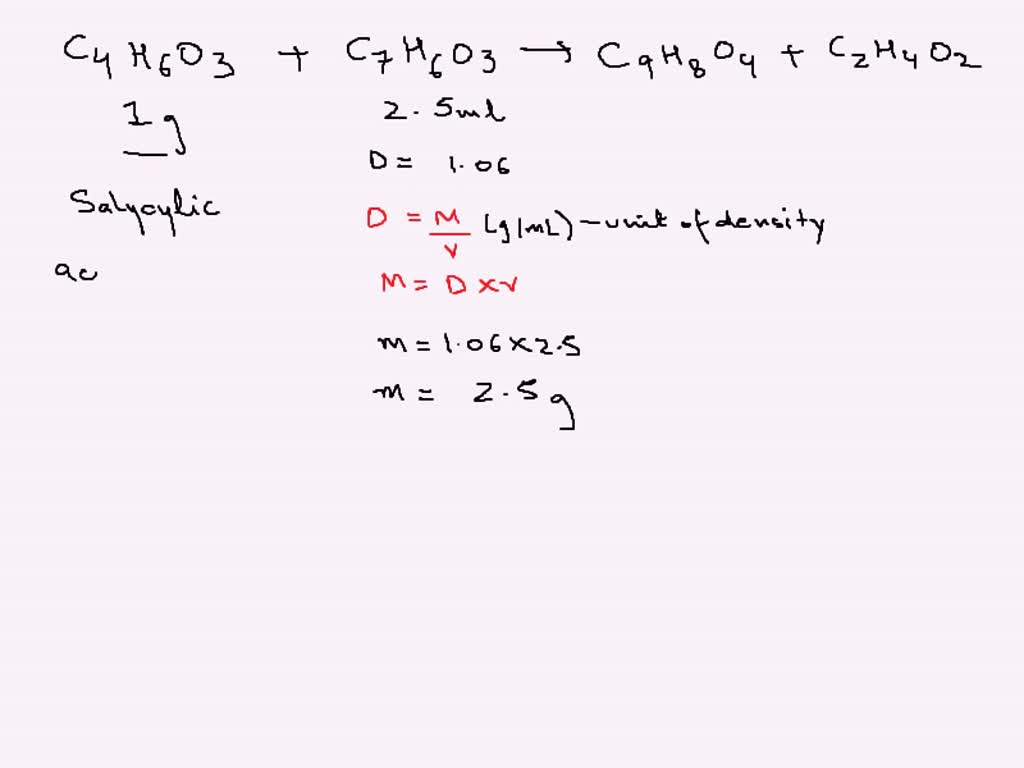
SOLVED: Calculate the theoretical yield of aspirin (C9H8O4) that would be obtained if 1.0 g of salicylic acid and 2.5 mL of acetic anhydride (density = 1.08 g/mL) were used as reactants.

SOLVED: Actual yield Yield % X [00 Theoretical yield starting materiul wtin grams MW of procuct in grams Theoretical Yicld MW of Starting Mat MW of nitrobenzaldehyde= 151 g/mole MW of Chalcone

Question Video: Calculating the Percentage Yield of the Recreation of Aqueous Copper Sulfate with Zinc Metal | Nagwa

SOLVED: Calculations to Show Theoretical yield of aspirin: Theoretical yield= no CzHsO CgHz CgHeS mass salicylic acid used" ( C-Hso mol C-HsO3' mol CgHa04 *#The molar masses of the salicylic acid and