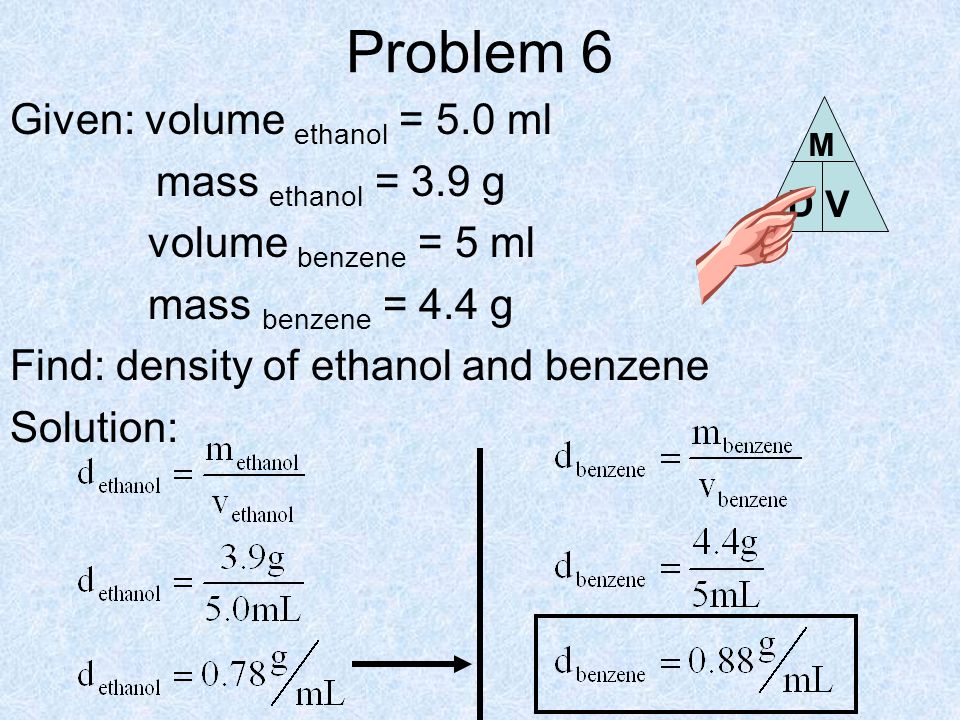
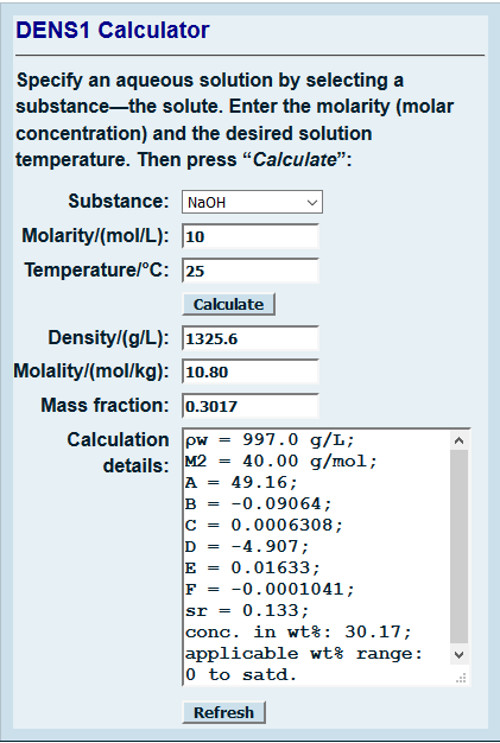
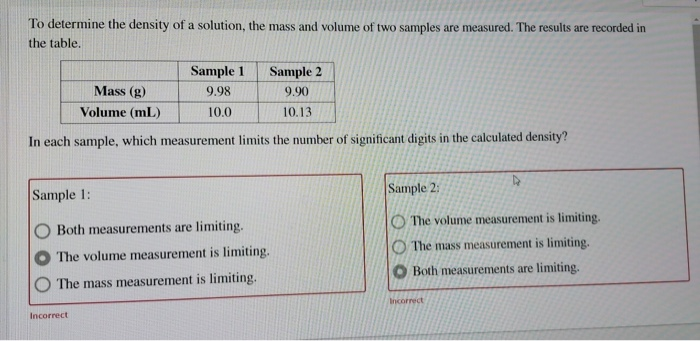
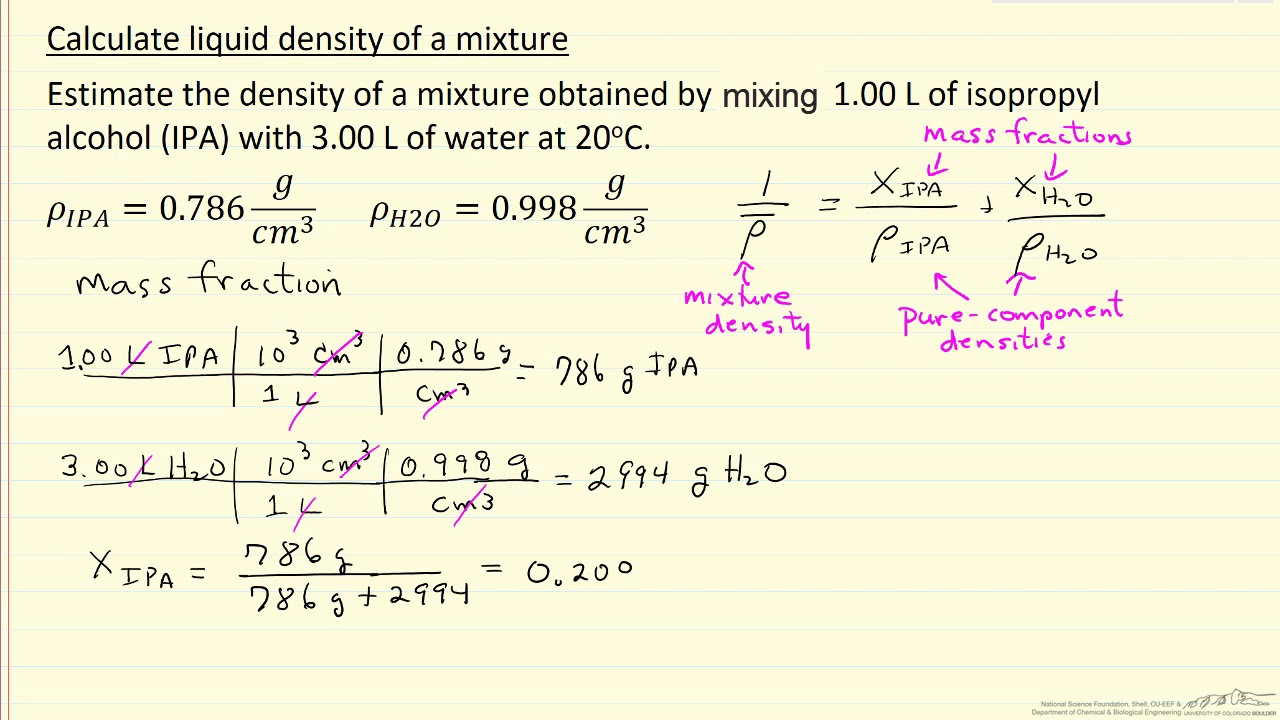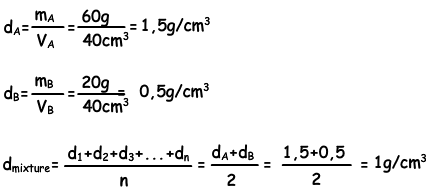SOLVED: To determine the density of solution; the mass and volume of two samples are measured. The results are recorded in the table Sample 9.97 10.0 Sample 2 9.80 10.19 Mass (g)

The density of 3M solution of NaCl is 1.25 g/mL. Calculate molality of the solution - CBSE Class 11 - Learn CBSE Forum

Molarity, Molality, Volume & Mass Percent, Mole Fraction & Density - Solution Concentration Problems - YouTube

Calculate the relative density of the liquid from the following information :Mass of empty relative density bottle = 24.5 g Mass of relative density bottle filled with water = 56.2 g Mass

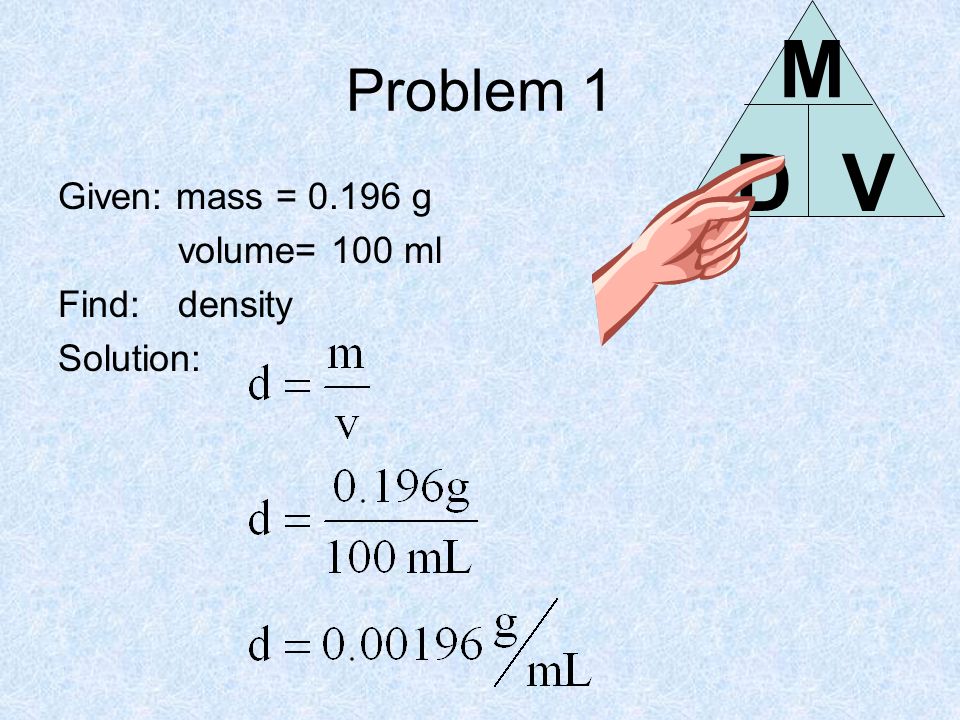
SOLVED: In order to calculate the density of a solution, you divide the mass of a liquid (5.10 g) by its volume (250.0 mL). How should you report its density?

:max_bytes(150000):strip_icc()/density-tower-showing-vase-with-5-layers-761602233-5a280d27842b170019ae91c3.jpg)



![Pharma Engineering: [HOW TO] Calculate Density of solvent Mixture Pharma Engineering: [HOW TO] Calculate Density of solvent Mixture](https://2.bp.blogspot.com/-IMT9YlNZa-M/V3ROrtEm7UI/AAAAAAAAAH0/vcCuiz3jQ0MyQfgXYyrDb2TfMC7SARykACLcB/w1200-h630-p-k-no-nu/slide_2.jpg)


![Pharma Engineering: [HOW TO] Calculate Density of solvent Mixture Pharma Engineering: [HOW TO] Calculate Density of solvent Mixture](https://2.bp.blogspot.com/-IMT9YlNZa-M/V3ROrtEm7UI/AAAAAAAAAH0/vcCuiz3jQ0MyQfgXYyrDb2TfMC7SARykACLcB/s1600/slide_2.jpg)