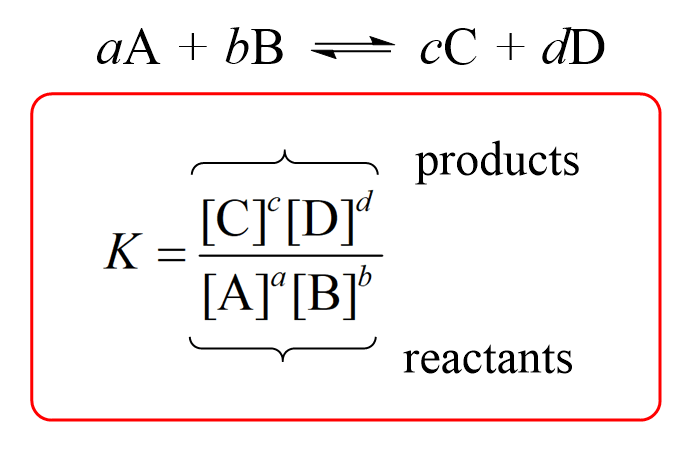
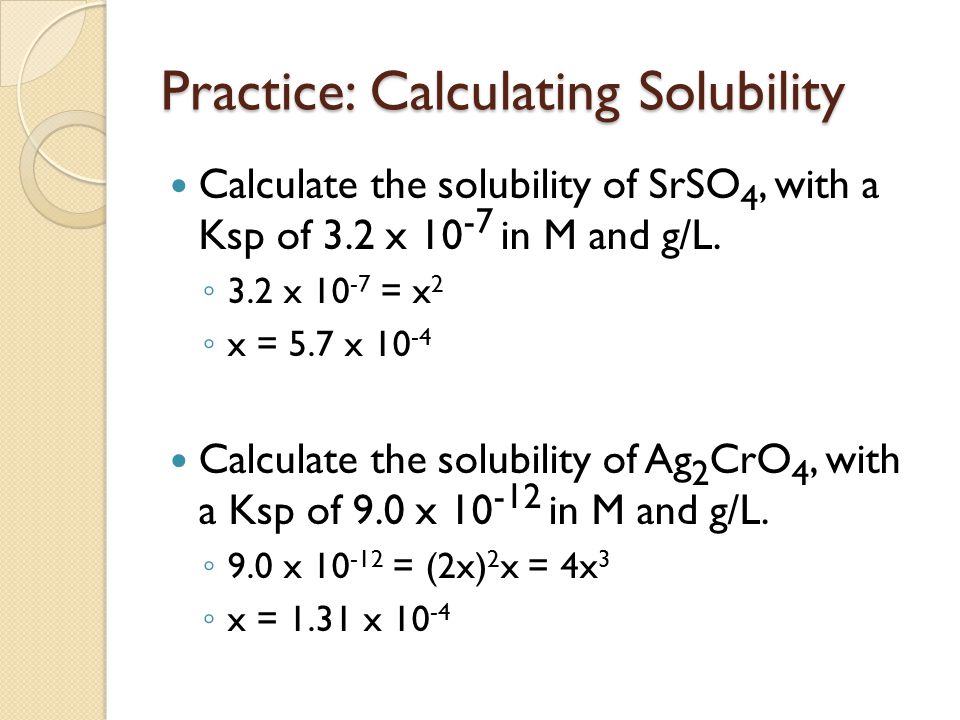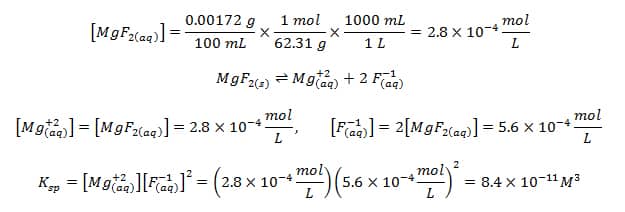Calculate the solubility of lead chloride in water, if its solubility product is 1.7 × 10^-5 .(Pb = 206, Cl = 35.5)
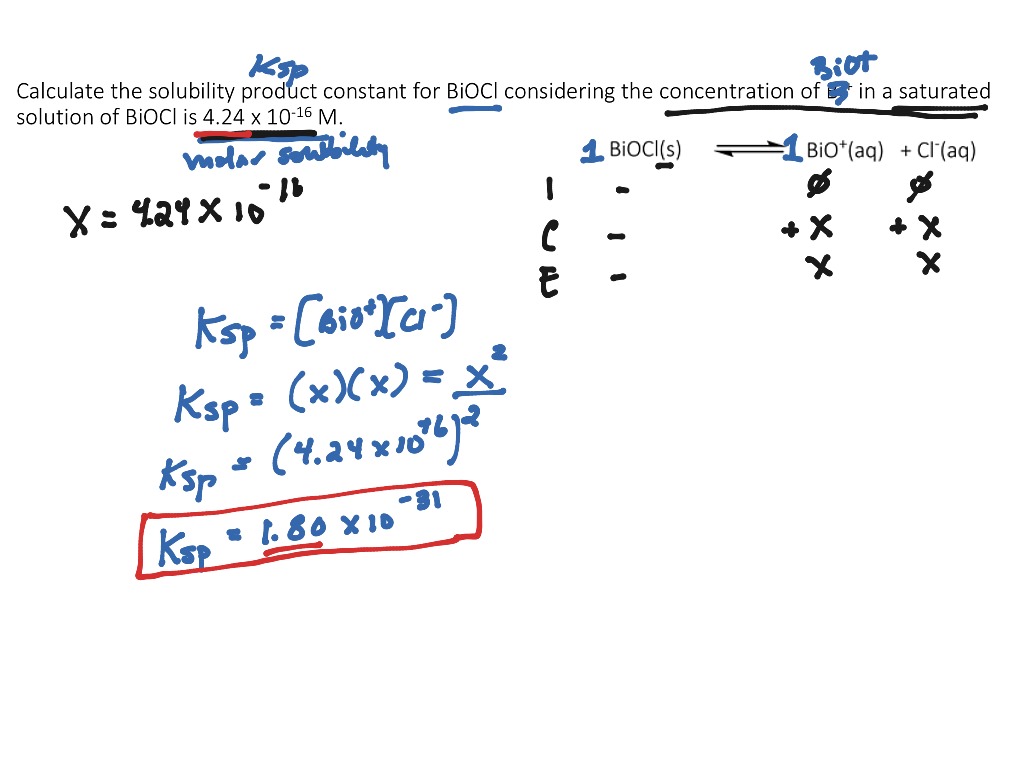
Calculating the Solubility Product Constant (Ksp) for an Insoluble Salt | Science, Chemistry | ShowMe
calculate solubility of silver benzoate in a buffer solution of ph=3.19 the ionisation cons†an t of benzoic acid is 6.46∗10^ 5 and Ksp for silver benzoate is 2.5∗10^ 13 (b) also calculate