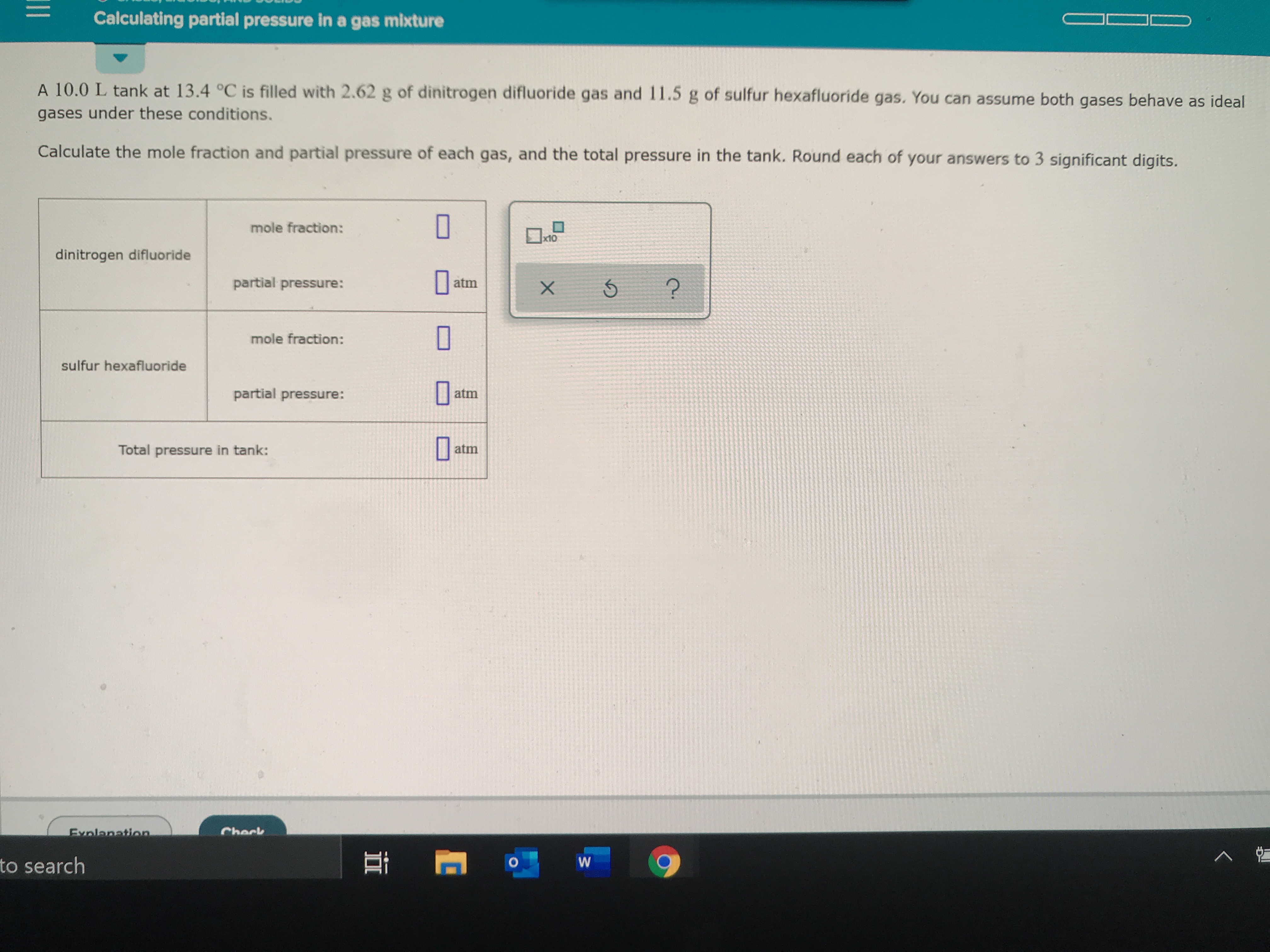
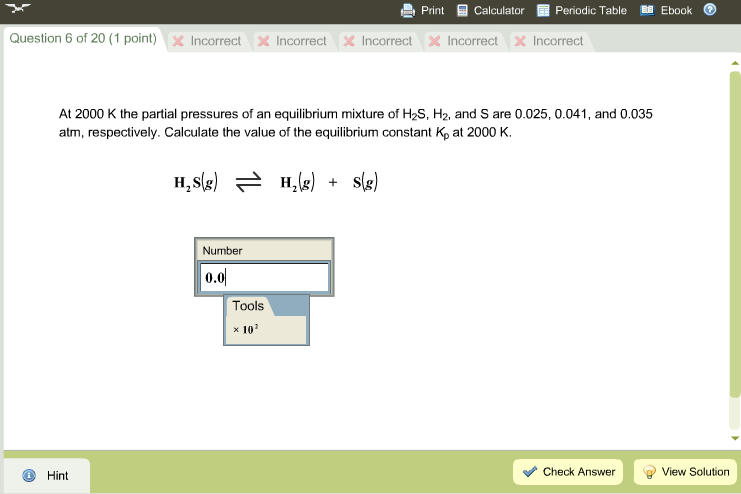
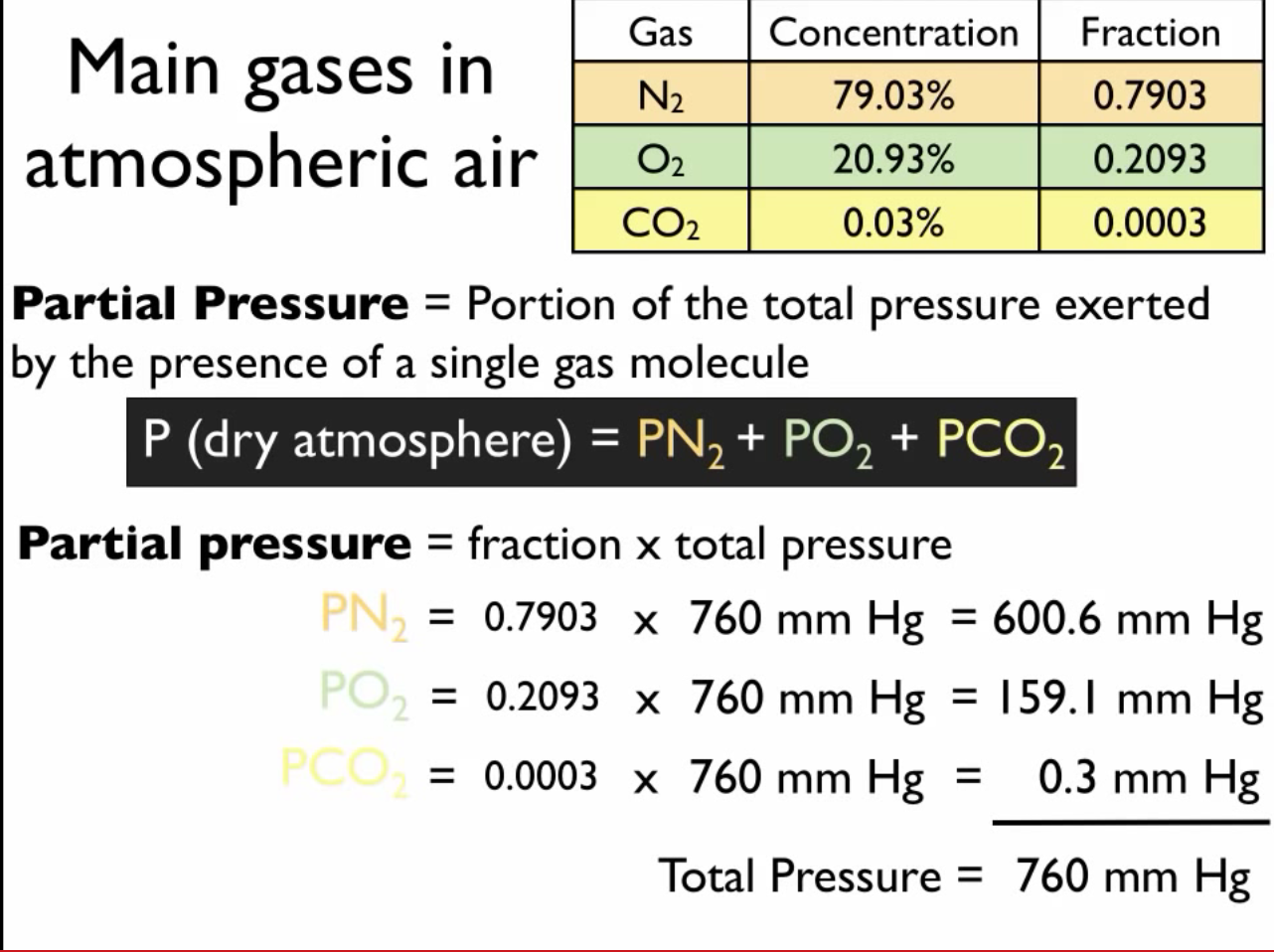
At 87^(@)C , the following equilibrium is established H(2) (g)+S(s)hArrH(2)S(g), K(p)=7xx10^(-2) If 0.50 mole of hydrogen and 1.0 mole of sulphur are heated to 87^(@)C calculate partial pressure of H2S at eq.

H2S , a toxic gas with a rotten egg - like smell, is used for the qualitative analysis. If the solubility of H2S in the water at STP is 0.195 m, then
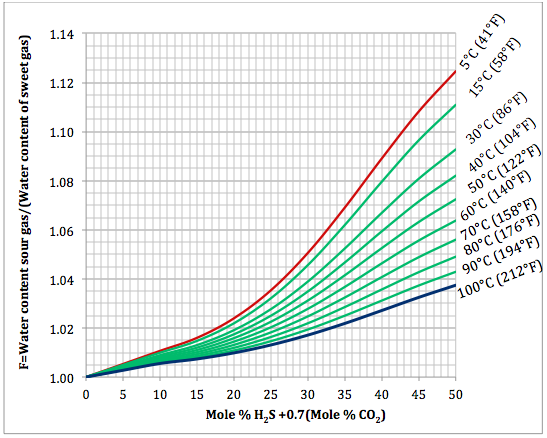
Estimating Sour Gas Water Content by New Correlations and Simplified Charts | Campbell Tip of the Month

Ammonium hydrogen sulphide dissociates as follows NH4HS (s) NH3 (g) + H2S (g) If solid NH4HS is placed in an evacuated flask at certain temperature it will dissociate until the total pressure