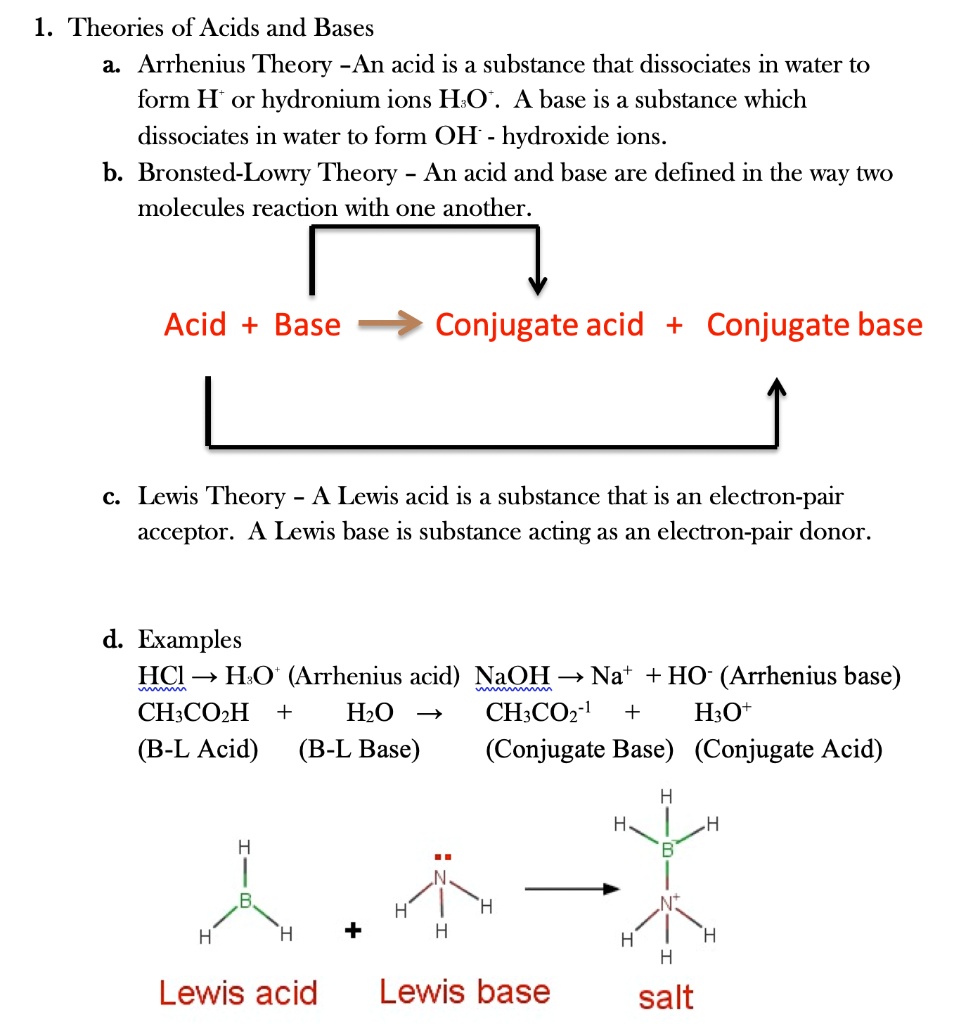
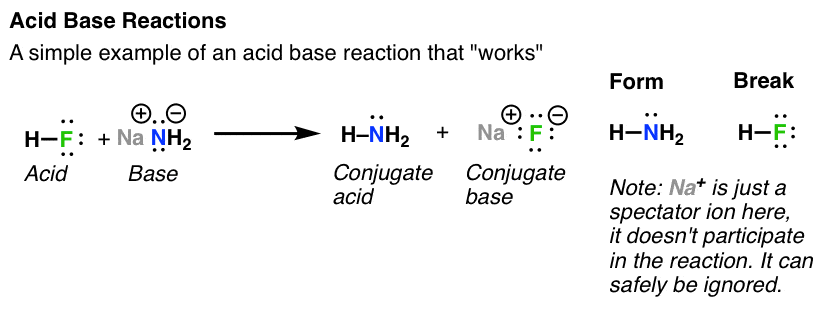
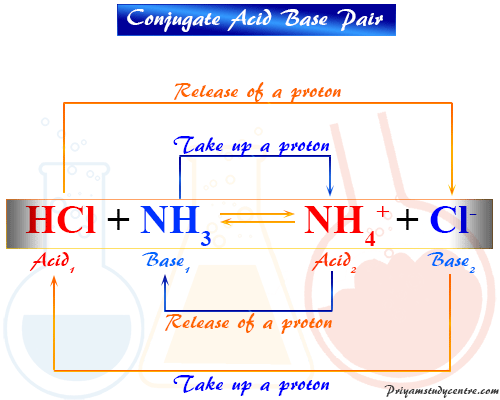
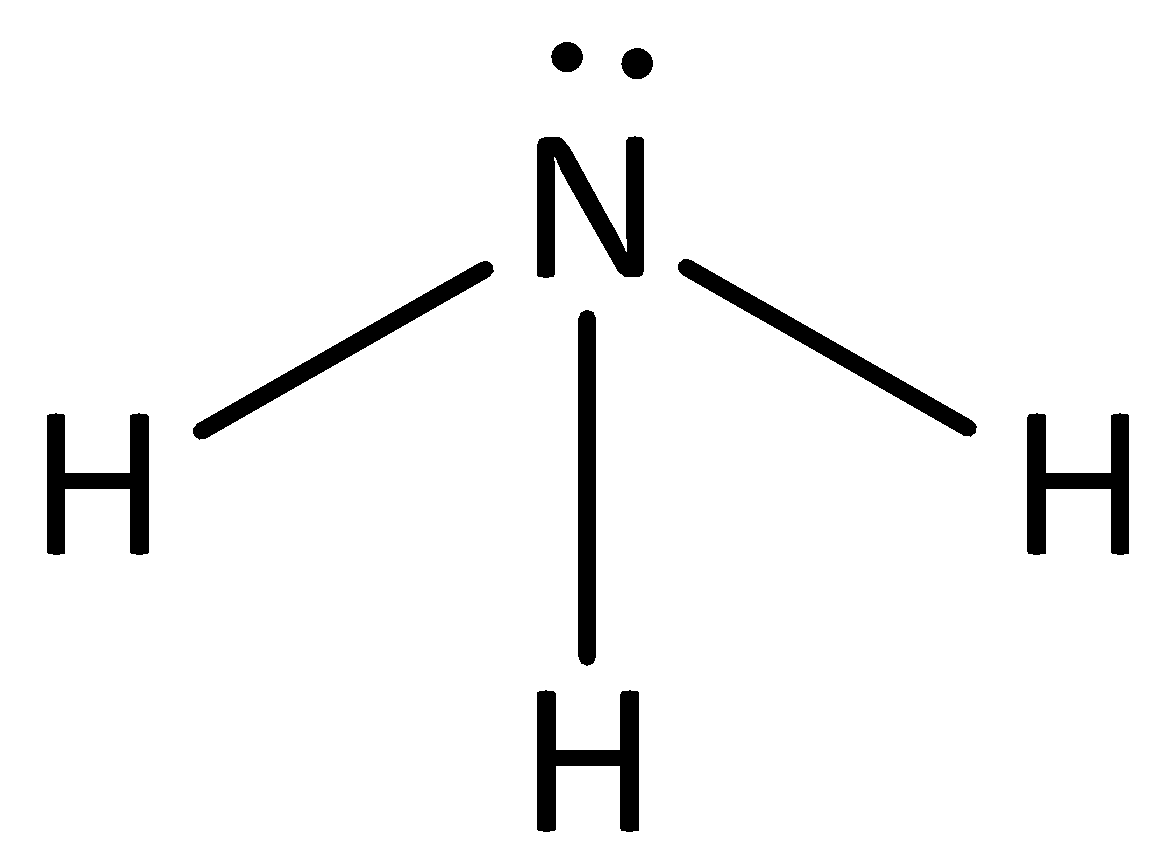
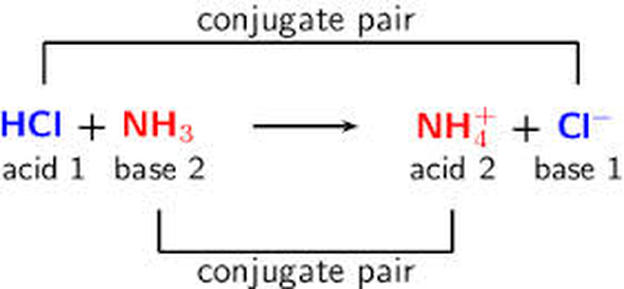
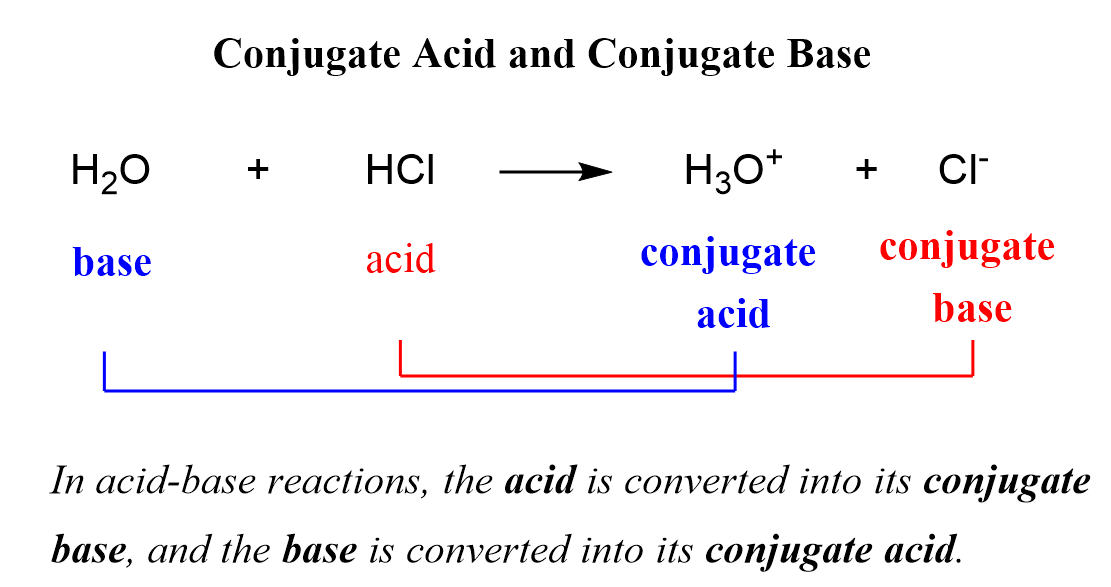
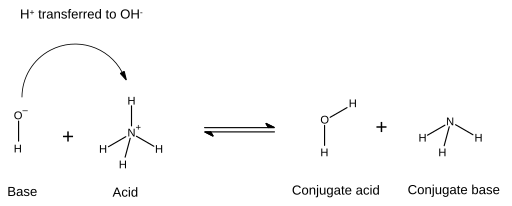
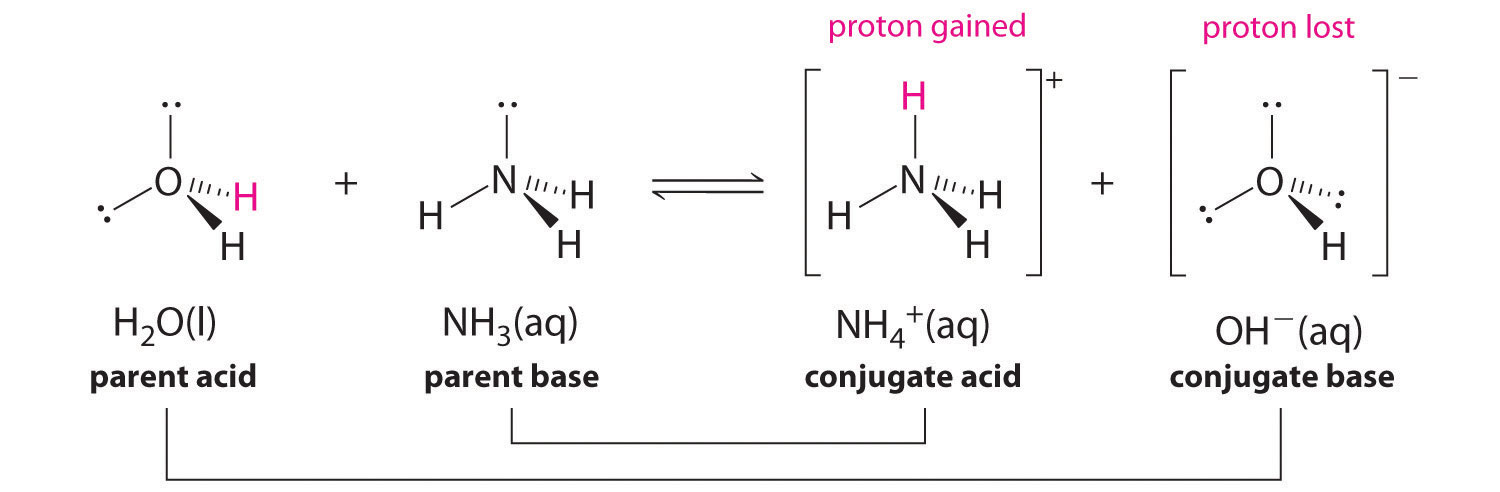
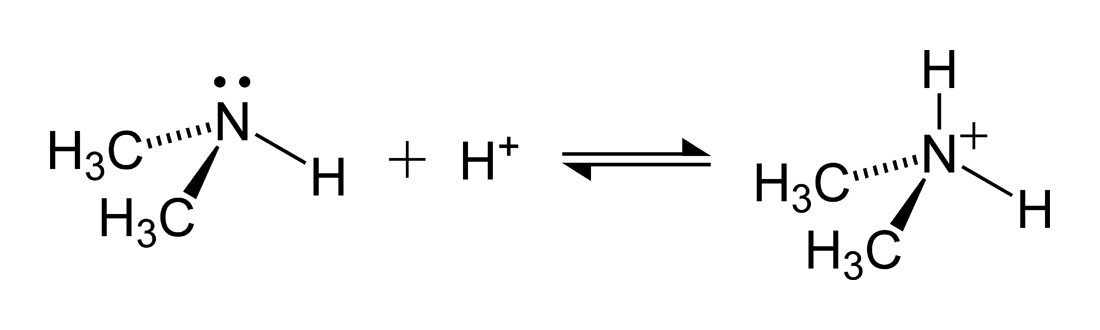![Chapter [ ] Acids and Bases Equilibria. Arrhenius (or Classical) Acid-Base Definition An acid is a substance that contains hydrogen and dissociates. - ppt download Chapter [ ] Acids and Bases Equilibria. Arrhenius (or Classical) Acid-Base Definition An acid is a substance that contains hydrogen and dissociates. - ppt download](https://images.slideplayer.com/23/6591516/slides/slide_9.jpg)
Chapter [ ] Acids and Bases Equilibria. Arrhenius (or Classical) Acid-Base Definition An acid is a substance that contains hydrogen and dissociates. - ppt download

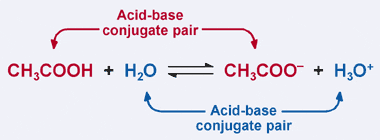
How to Identify Acid, Base, Conjugate Acid, and Conjugate Base Examples and Practice Problems - YouTube