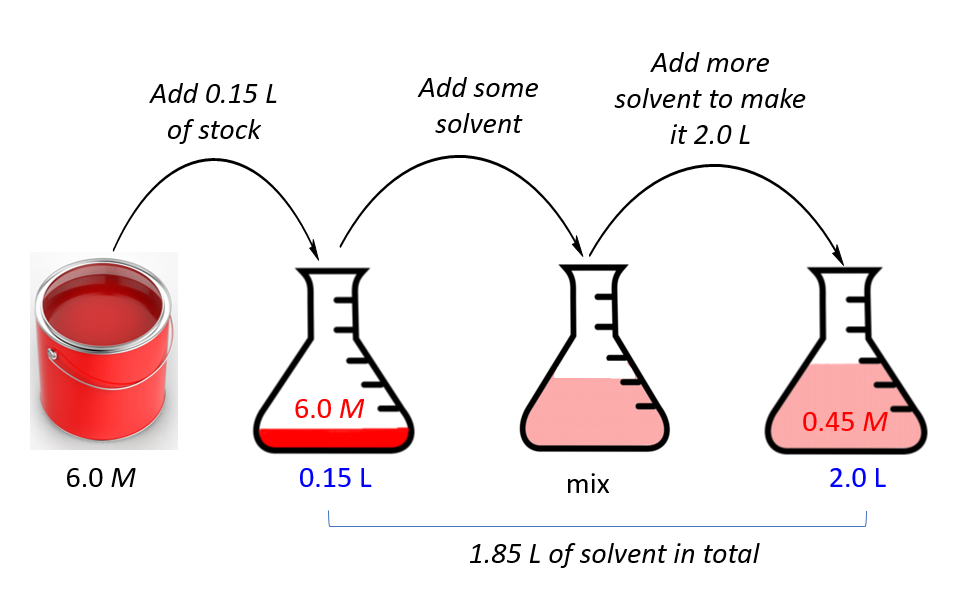
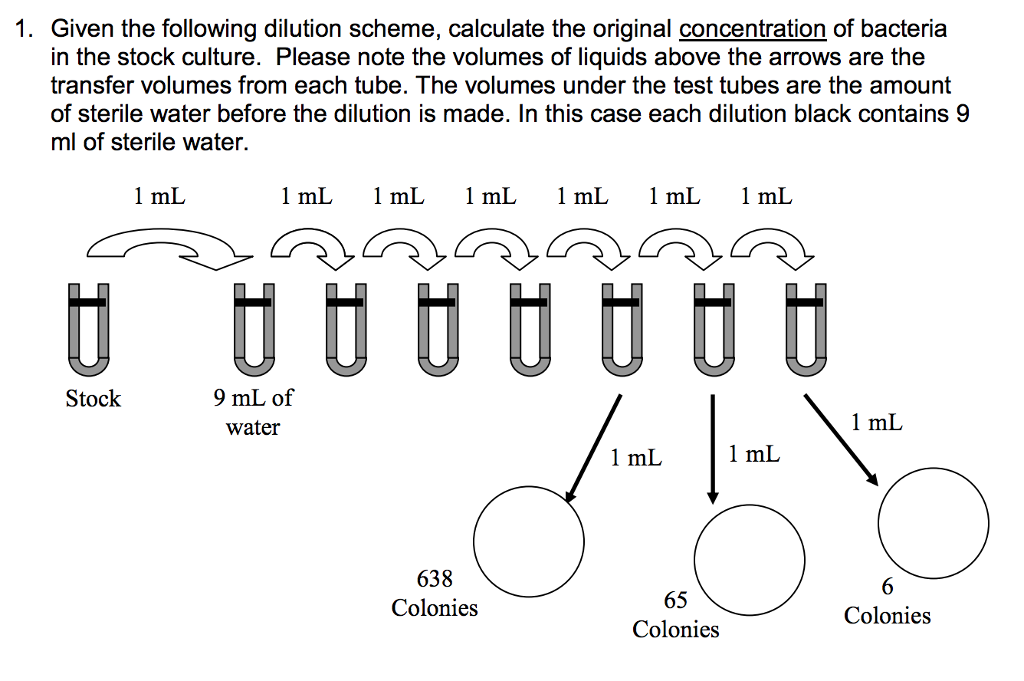
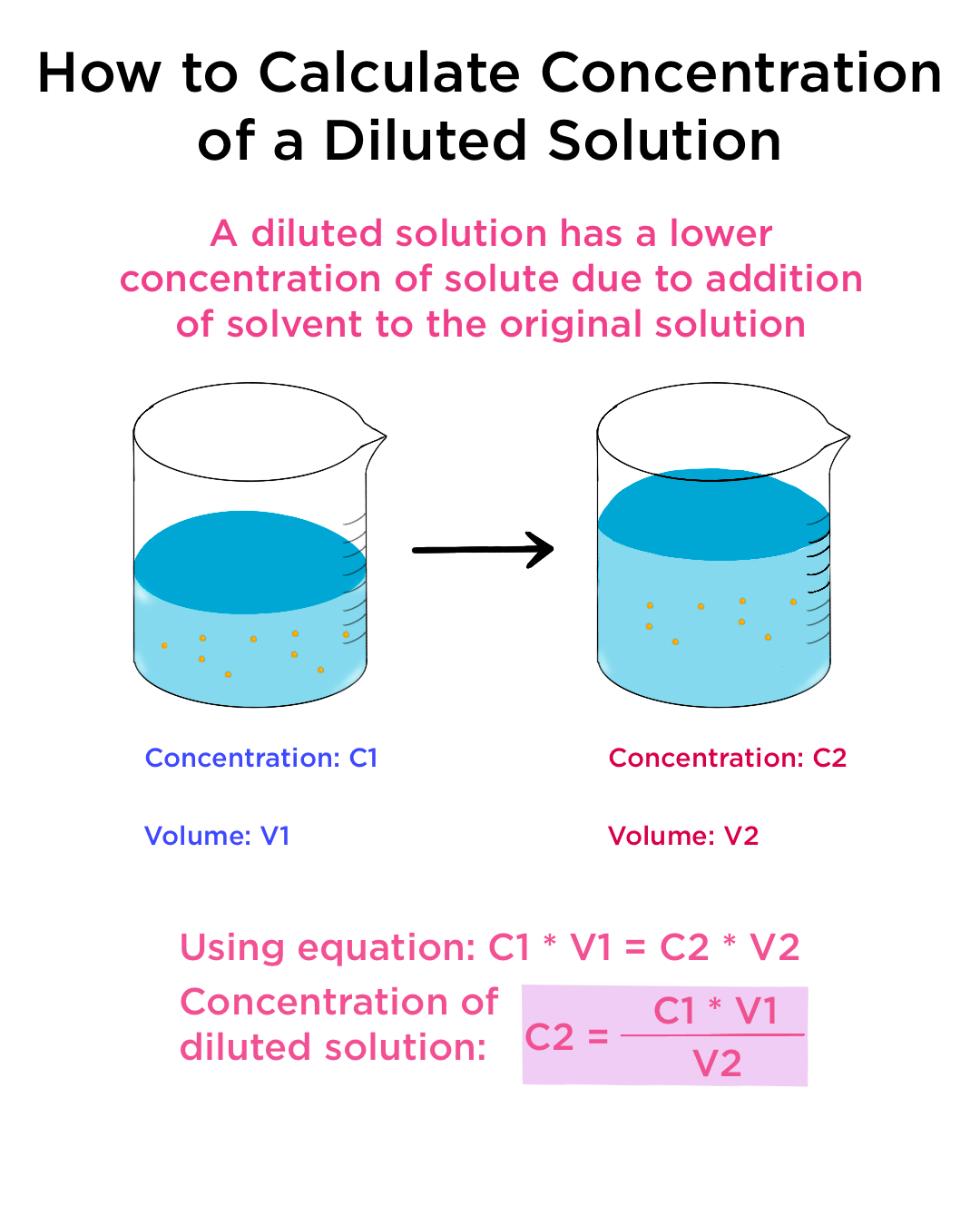
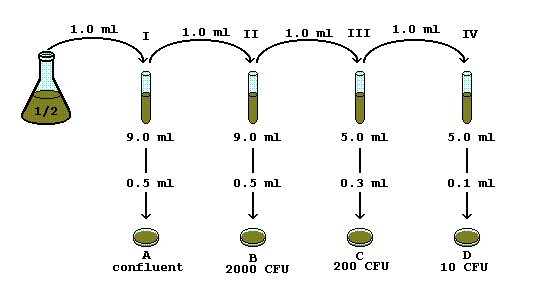
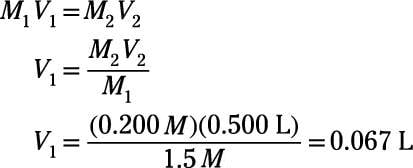SOLVED: DILUTION AND CONCENTRATION When liquid medication of a given strength is diluted, its strength will be reduced. For example, 10 mL of a solution containing g of a substance has strength

Dilution Equation & Examples | How to Calculate Dilution Factors - Video & Lesson Transcript | Study.com

1 Chapter 10 Acids and Bases 10.10Dilutions. 2 Dilution Diluting a solution Is the addition of water. Decreases concentration. ConcentratedDilutedSolution. - ppt download
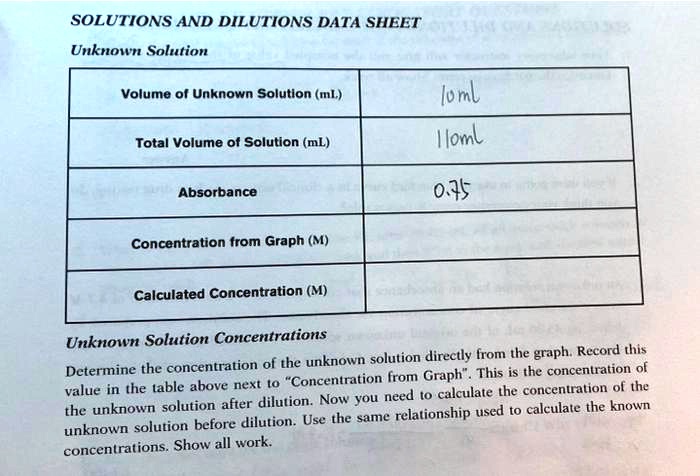
SOLVED: SOLUTIONS AND DILUTIONS DATA SHEET Unknown Solution Volume ol Unknown Solutlon (ml;) loml Iloml Total Volume ot Solution (mL) Absorbance 0.35 Concentration from Graph (M) Calculated Concentration (M) Unknown Solution Concentrations