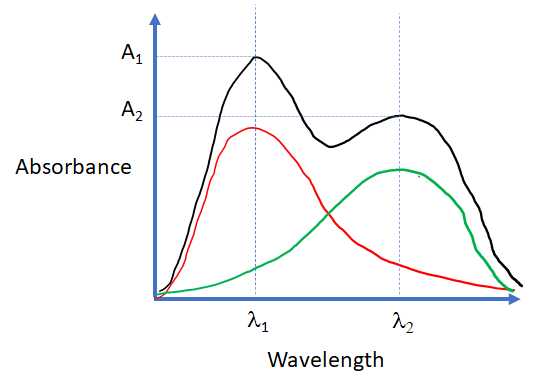
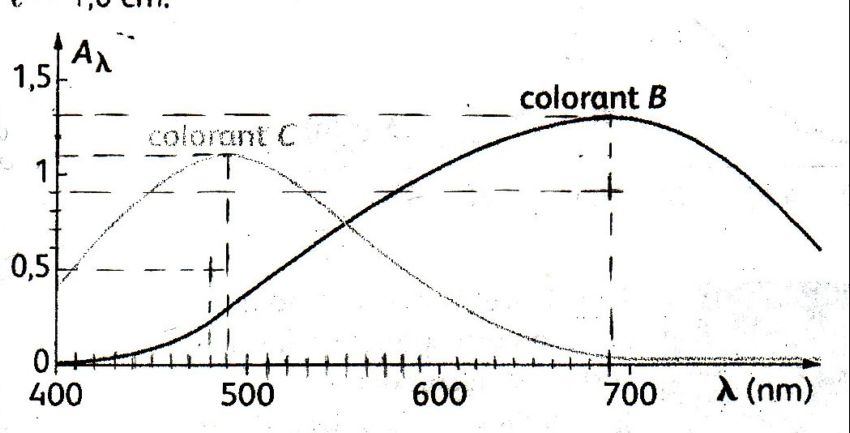
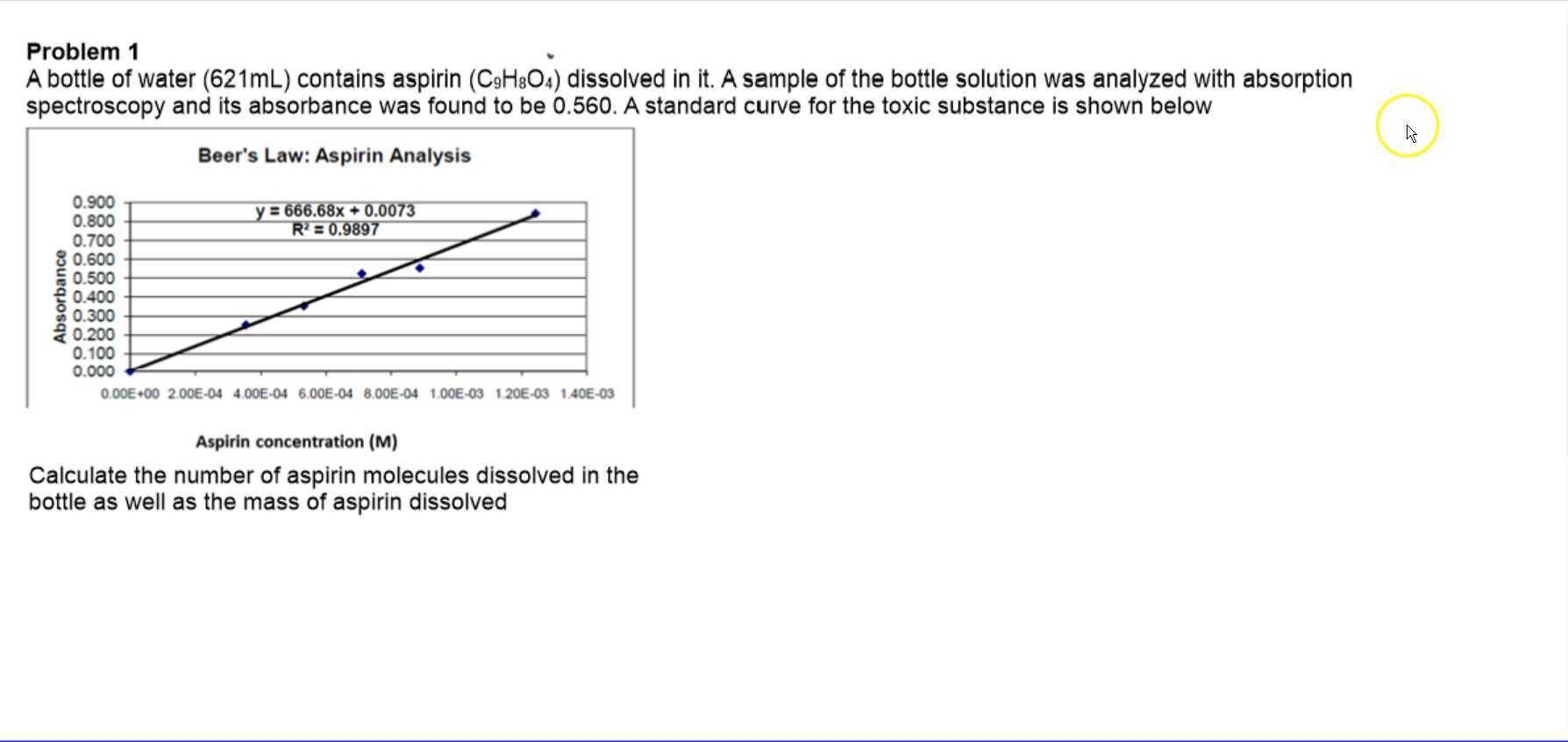
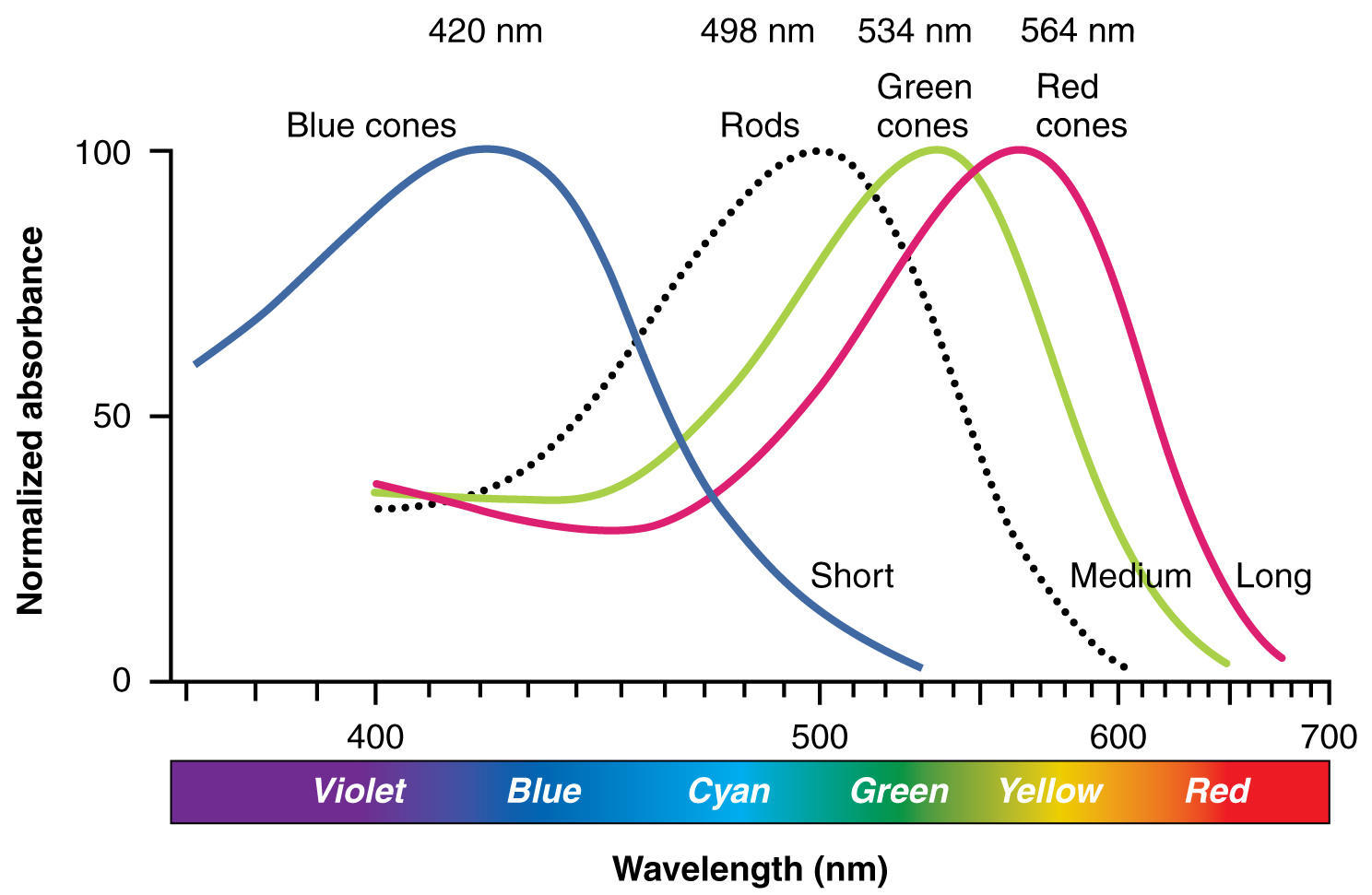
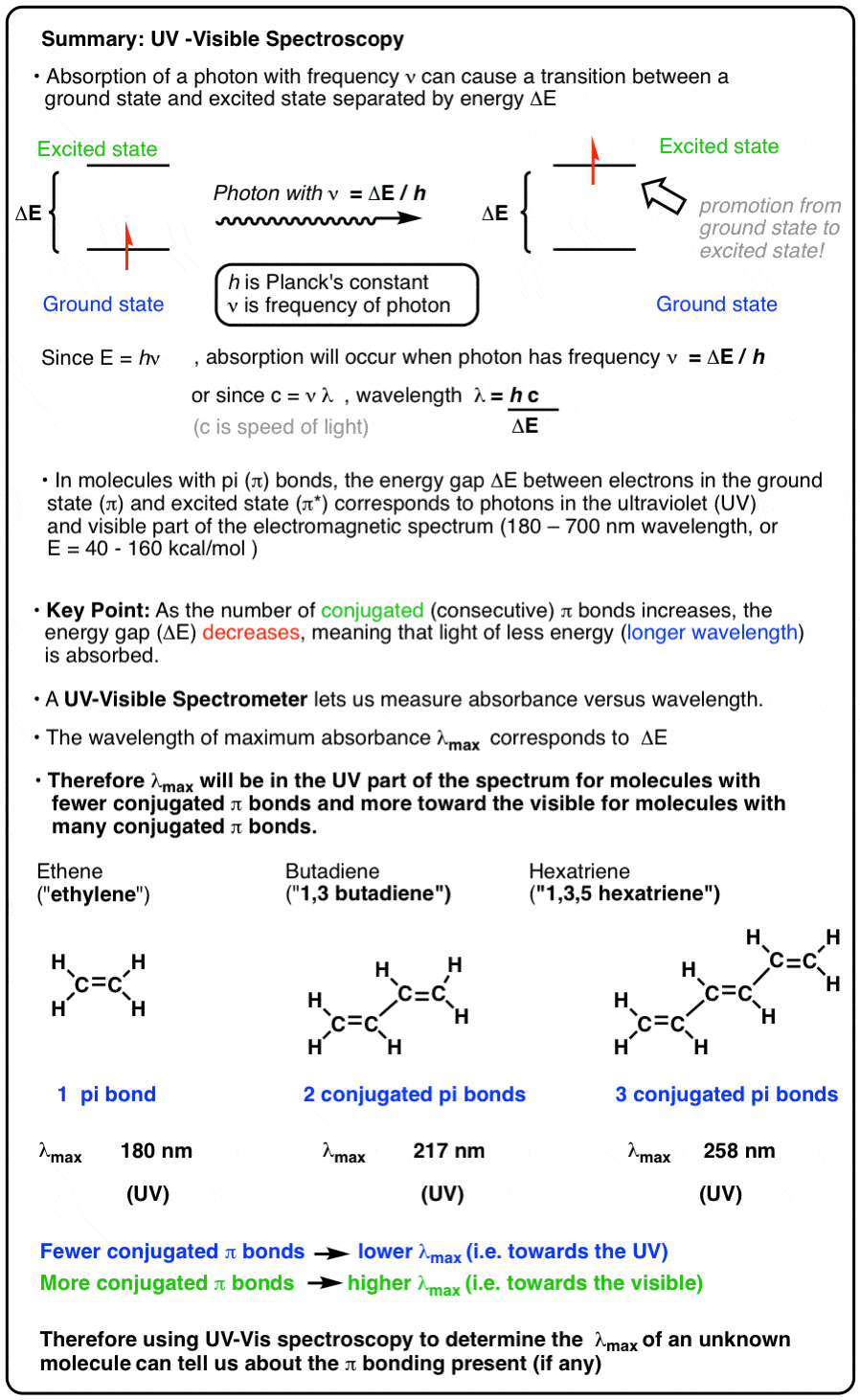
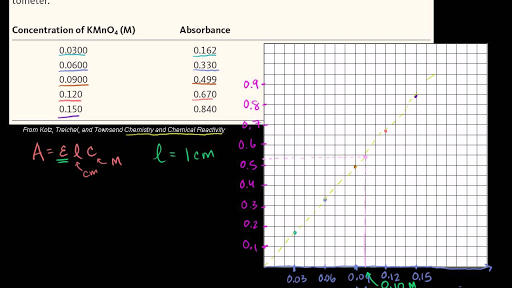
SOLVED: Absorption Max Wavelength (nm) 309 IQQUM Absorbance 2.29 5QuM Absorbance 1.13 25UM Absorbance 0.54 Unknown Absorbance 1.71 Slope 23.314 y-intercept Unknown EDMAB Concentration 0.0733 Using your value for molar absorptivity at

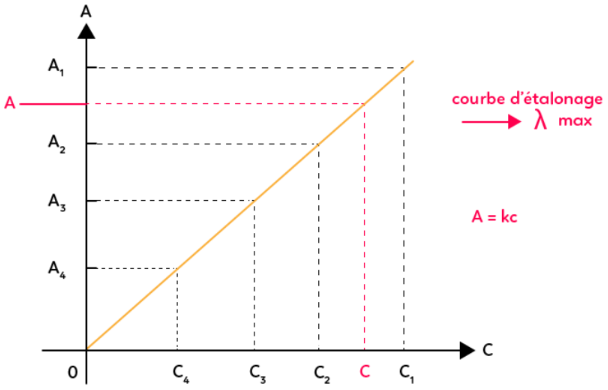
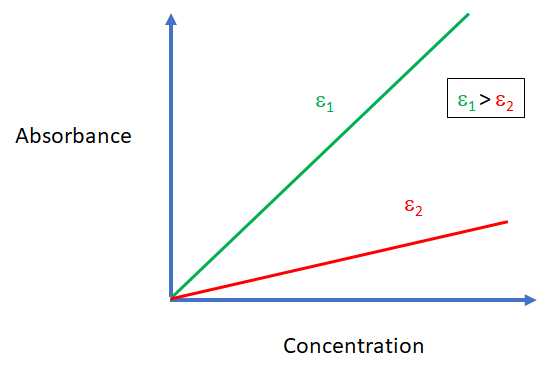
SOLVED: Q4: Beer-Lambert law states that absorbance of the sample is directly proportional to concentration A= €. C. A: Absorbance €: Molar absorptivity coefficient (M-1 cm-1) C: Concentration L: Path length (usually

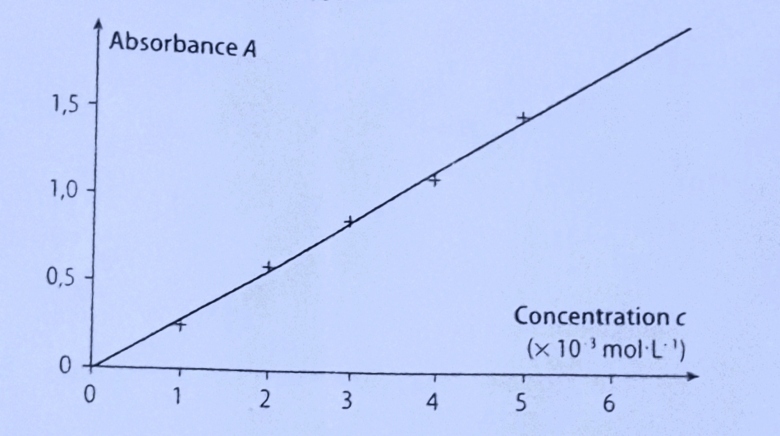
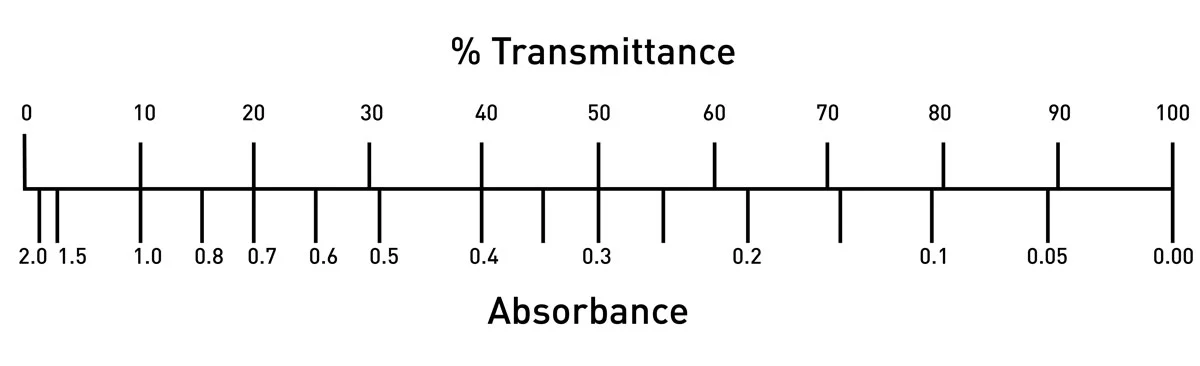
Plot an absorbance vs concentration and %T vs concentration in one graph? (The absorbance values are so small compare to the %T values.) | Homework.Study.com
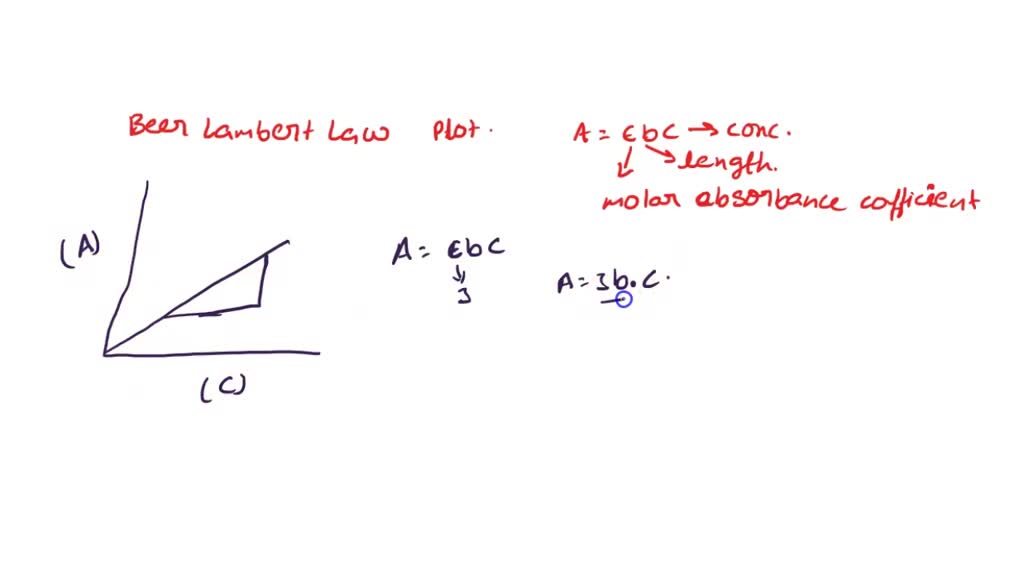
SOLVED: In the Beer-Lambert law plot, what does the x-axis represent from the absorbance equation (a=3bc)? What does the y-axis represent? In the equation for the straight line for the Beer-Lampert plot (