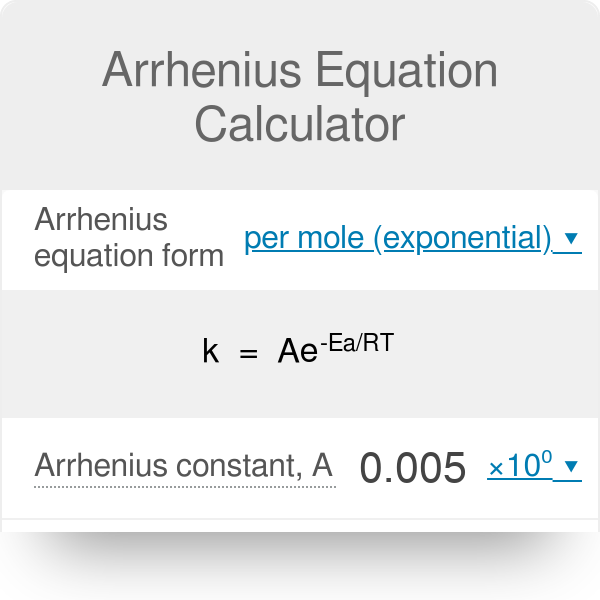
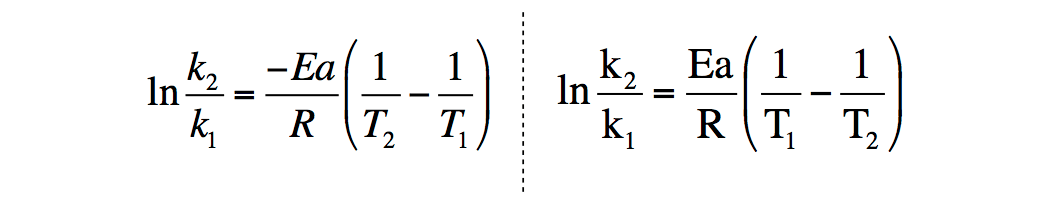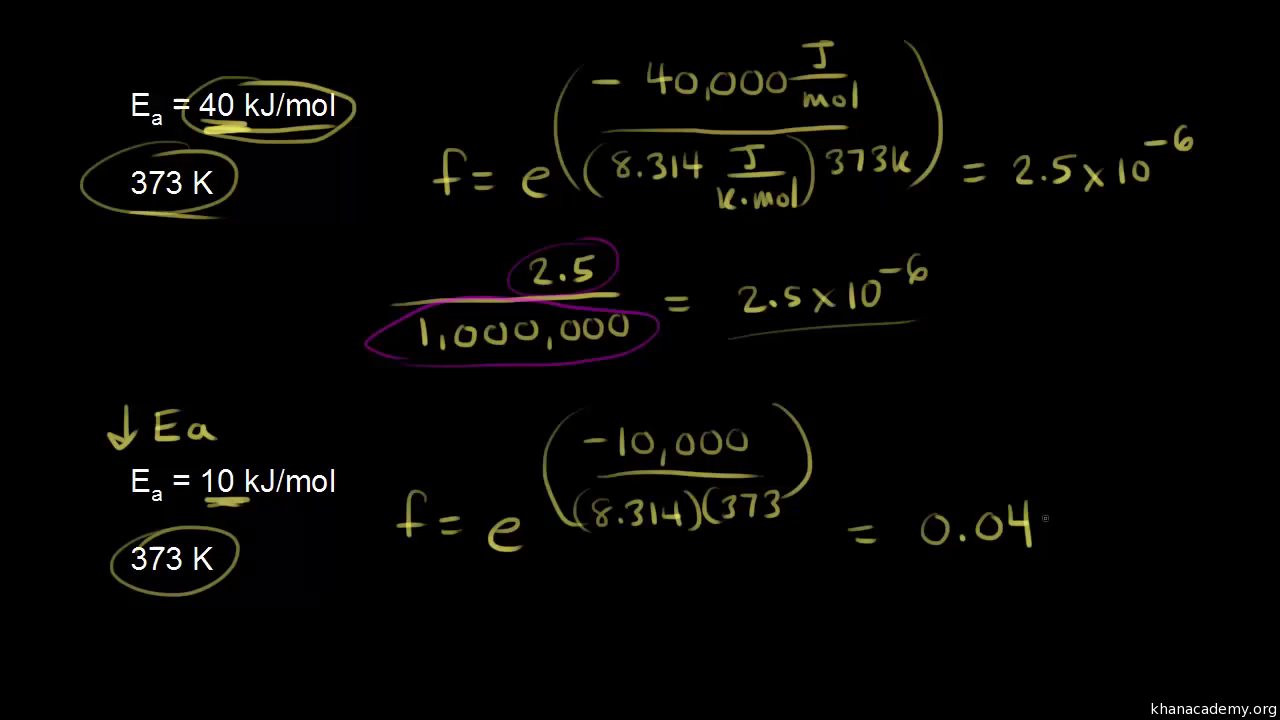Is there a way to calculate values in the Arrhenius equation without a graphing calculator? | Socratic

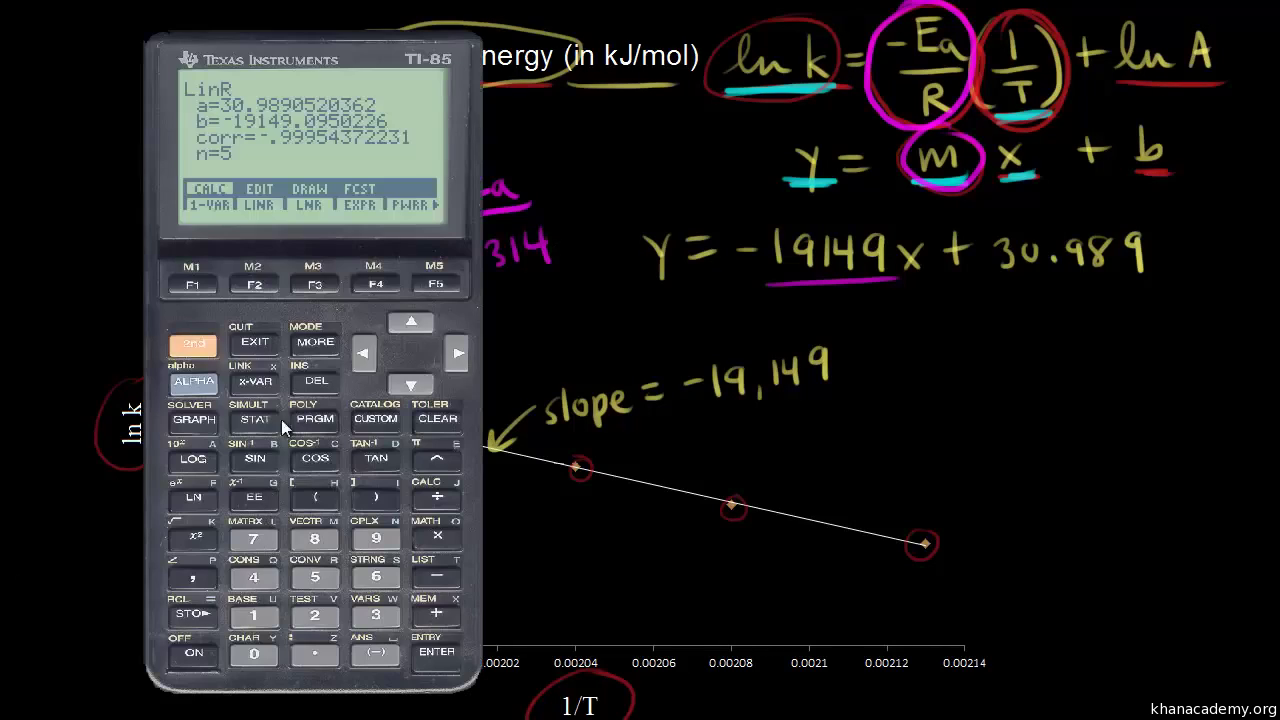
Arrhenius equation LO- Carry out calculations involving the Arrhenius equation. So far we have looked quantitatively at how to show the effect of a concentration. - ppt download

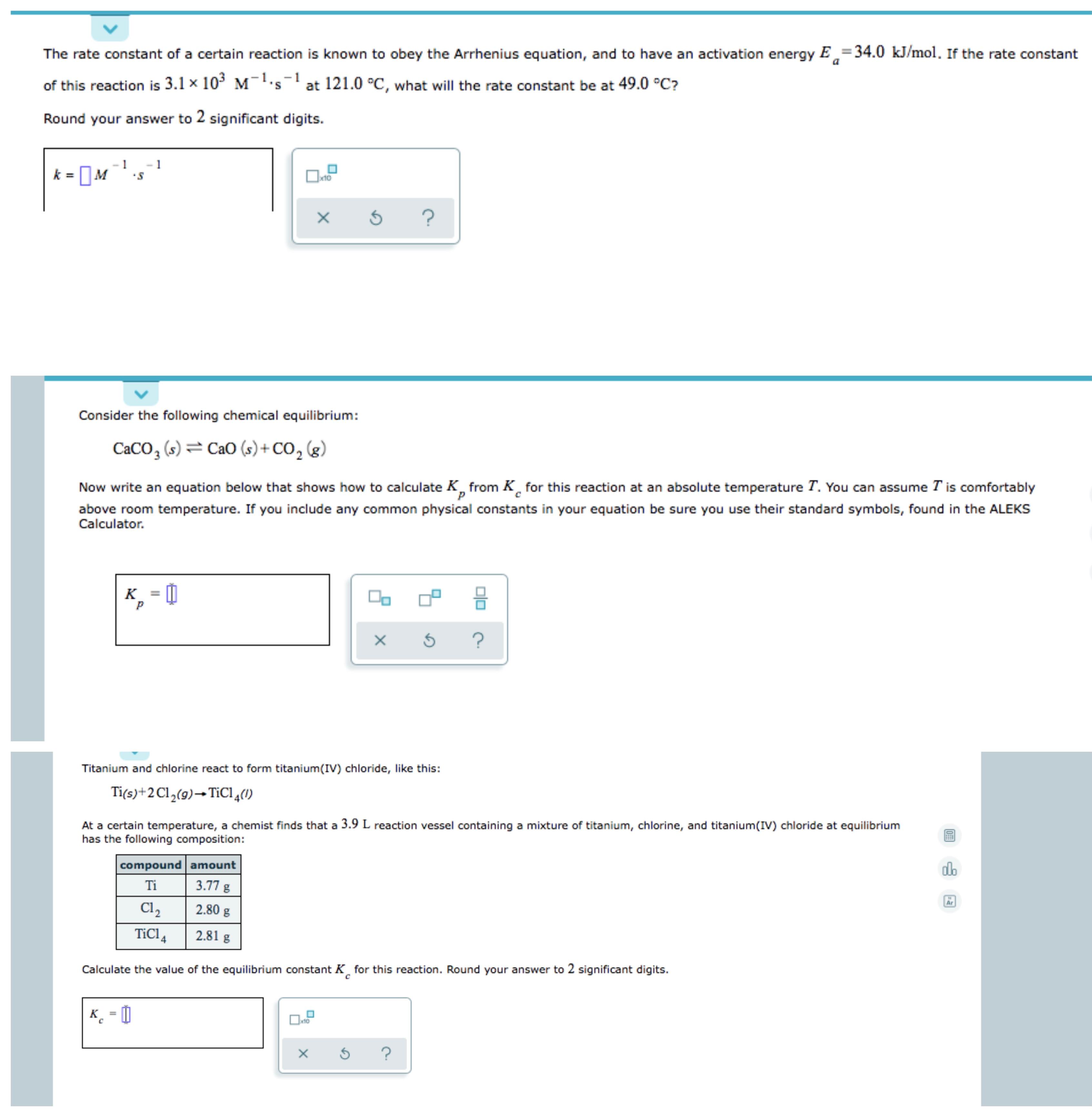
SOLVED: The rate constant k for a certain reaction is measured at two different temperatures: temperature 91.0 'C 4.1x 10 31.0 ' 1.7* 10 Assuming the rate constant obeys the Arrhenius equation,