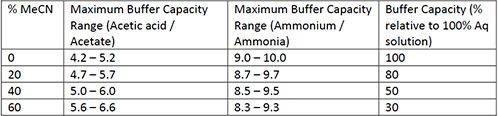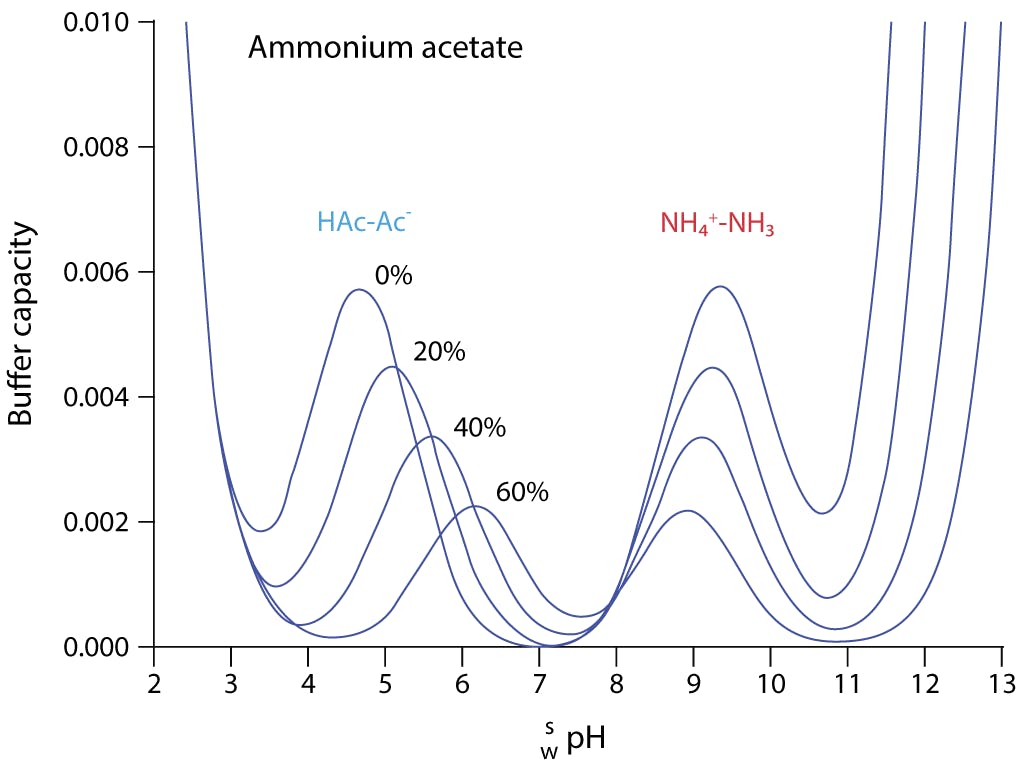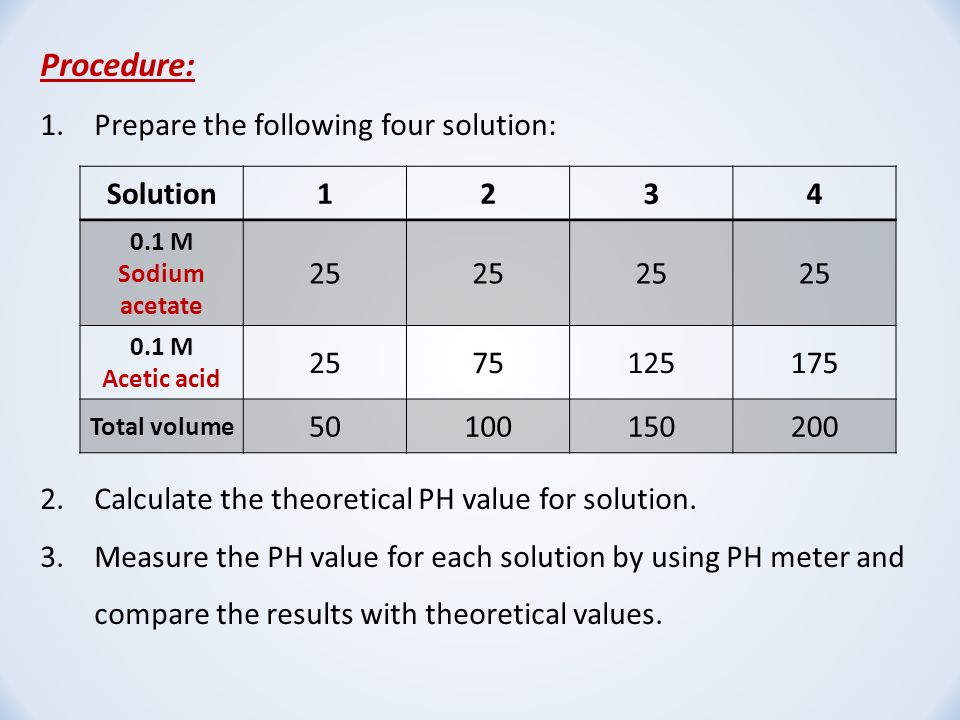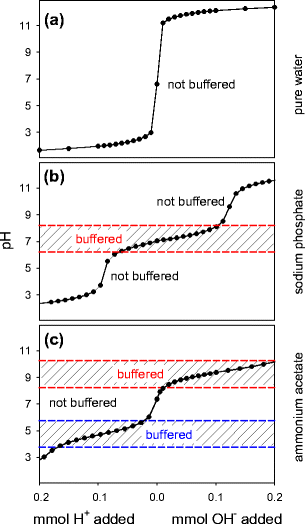
Addressing a Common Misconception: Ammonium Acetate as Neutral pH “Buffer” for Native Electrospray Mass Spectrometry | SpringerLink

Calculated buffer capacity at different pH for a) 0.10 M acetate and b)... | Download Scientific Diagram

Ammonium acetate (C<sub>2</sub>H<sub>7</sub>NO<sub>2</sub>) - Structure, properties , Production, Uses and FAQs of Ammonium acetate.

A buffer solution can be prepared from a mixture of:(i) Sodium acetate and acetic acid in water(ii) Sodium acetate and hydrochloric acid in water(iii) Ammonia and ammonium chloride in water(iv) Ammonia and

Breaking old habits: Moving away from commonly used buffers in pharmaceuticals - European Pharmaceutical Review

![Ammonium Acetate [CH3COONH4] Molecular Weight Calculation - Laboratory Notes Ammonium Acetate [CH3COONH4] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/03/ammonium-acetate-molecular-weight-calculation-300x200.jpg)